An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.

National Institute of Environmental Health Sciences
Your environment. your health., what is ethics in research & why is it important, by david b. resnik, j.d., ph.d..
December 23, 2020
The ideas and opinions expressed in this essay are the author’s own and do not necessarily represent those of the NIH, NIEHS, or US government.

When most people think of ethics (or morals), they think of rules for distinguishing between right and wrong, such as the Golden Rule ("Do unto others as you would have them do unto you"), a code of professional conduct like the Hippocratic Oath ("First of all, do no harm"), a religious creed like the Ten Commandments ("Thou Shalt not kill..."), or a wise aphorisms like the sayings of Confucius. This is the most common way of defining "ethics": norms for conduct that distinguish between acceptable and unacceptable behavior.
Most people learn ethical norms at home, at school, in church, or in other social settings. Although most people acquire their sense of right and wrong during childhood, moral development occurs throughout life and human beings pass through different stages of growth as they mature. Ethical norms are so ubiquitous that one might be tempted to regard them as simple commonsense. On the other hand, if morality were nothing more than commonsense, then why are there so many ethical disputes and issues in our society?
Alternatives to Animal Testing
Alternative test methods are methods that replace, reduce, or refine animal use in research and testing
Learn more about Environmental science Basics
One plausible explanation of these disagreements is that all people recognize some common ethical norms but interpret, apply, and balance them in different ways in light of their own values and life experiences. For example, two people could agree that murder is wrong but disagree about the morality of abortion because they have different understandings of what it means to be a human being.
Most societies also have legal rules that govern behavior, but ethical norms tend to be broader and more informal than laws. Although most societies use laws to enforce widely accepted moral standards and ethical and legal rules use similar concepts, ethics and law are not the same. An action may be legal but unethical or illegal but ethical. We can also use ethical concepts and principles to criticize, evaluate, propose, or interpret laws. Indeed, in the last century, many social reformers have urged citizens to disobey laws they regarded as immoral or unjust laws. Peaceful civil disobedience is an ethical way of protesting laws or expressing political viewpoints.
Another way of defining 'ethics' focuses on the disciplines that study standards of conduct, such as philosophy, theology, law, psychology, or sociology. For example, a "medical ethicist" is someone who studies ethical standards in medicine. One may also define ethics as a method, procedure, or perspective for deciding how to act and for analyzing complex problems and issues. For instance, in considering a complex issue like global warming , one may take an economic, ecological, political, or ethical perspective on the problem. While an economist might examine the cost and benefits of various policies related to global warming, an environmental ethicist could examine the ethical values and principles at stake.
See ethics in practice at NIEHS
Read latest updates in our monthly Global Environmental Health Newsletter

Many different disciplines, institutions , and professions have standards for behavior that suit their particular aims and goals. These standards also help members of the discipline to coordinate their actions or activities and to establish the public's trust of the discipline. For instance, ethical standards govern conduct in medicine, law, engineering, and business. Ethical norms also serve the aims or goals of research and apply to people who conduct scientific research or other scholarly or creative activities. There is even a specialized discipline, research ethics, which studies these norms. See Glossary of Commonly Used Terms in Research Ethics and Research Ethics Timeline .
There are several reasons why it is important to adhere to ethical norms in research. First, norms promote the aims of research , such as knowledge, truth, and avoidance of error. For example, prohibitions against fabricating , falsifying, or misrepresenting research data promote the truth and minimize error.
Join an NIEHS Study
See how we put research Ethics to practice.
Visit Joinastudy.niehs.nih.gov to see the various studies NIEHS perform.

Second, since research often involves a great deal of cooperation and coordination among many different people in different disciplines and institutions, ethical standards promote the values that are essential to collaborative work , such as trust, accountability, mutual respect, and fairness. For example, many ethical norms in research, such as guidelines for authorship , copyright and patenting policies , data sharing policies, and confidentiality rules in peer review, are designed to protect intellectual property interests while encouraging collaboration. Most researchers want to receive credit for their contributions and do not want to have their ideas stolen or disclosed prematurely.
Third, many of the ethical norms help to ensure that researchers can be held accountable to the public . For instance, federal policies on research misconduct, conflicts of interest, the human subjects protections, and animal care and use are necessary in order to make sure that researchers who are funded by public money can be held accountable to the public.
Fourth, ethical norms in research also help to build public support for research. People are more likely to fund a research project if they can trust the quality and integrity of research.
Finally, many of the norms of research promote a variety of other important moral and social values , such as social responsibility, human rights, animal welfare, compliance with the law, and public health and safety. Ethical lapses in research can significantly harm human and animal subjects, students, and the public. For example, a researcher who fabricates data in a clinical trial may harm or even kill patients, and a researcher who fails to abide by regulations and guidelines relating to radiation or biological safety may jeopardize his health and safety or the health and safety of staff and students.
Codes and Policies for Research Ethics
Given the importance of ethics for the conduct of research, it should come as no surprise that many different professional associations, government agencies, and universities have adopted specific codes, rules, and policies relating to research ethics. Many government agencies have ethics rules for funded researchers.
- National Institutes of Health (NIH)
- National Science Foundation (NSF)
- Food and Drug Administration (FDA)
- Environmental Protection Agency (EPA)
- US Department of Agriculture (USDA)
- Singapore Statement on Research Integrity
- American Chemical Society, The Chemist Professional’s Code of Conduct
- Code of Ethics (American Society for Clinical Laboratory Science)
- American Psychological Association, Ethical Principles of Psychologists and Code of Conduct
- Statement on Professional Ethics (American Association of University Professors)
- Nuremberg Code
- World Medical Association's Declaration of Helsinki
Ethical Principles
The following is a rough and general summary of some ethical principles that various codes address*:

Strive for honesty in all scientific communications. Honestly report data, results, methods and procedures, and publication status. Do not fabricate, falsify, or misrepresent data. Do not deceive colleagues, research sponsors, or the public.

Objectivity
Strive to avoid bias in experimental design, data analysis, data interpretation, peer review, personnel decisions, grant writing, expert testimony, and other aspects of research where objectivity is expected or required. Avoid or minimize bias or self-deception. Disclose personal or financial interests that may affect research.

Keep your promises and agreements; act with sincerity; strive for consistency of thought and action.

Carefulness
Avoid careless errors and negligence; carefully and critically examine your own work and the work of your peers. Keep good records of research activities, such as data collection, research design, and correspondence with agencies or journals.

Share data, results, ideas, tools, resources. Be open to criticism and new ideas.

Transparency
Disclose methods, materials, assumptions, analyses, and other information needed to evaluate your research.

Accountability
Take responsibility for your part in research and be prepared to give an account (i.e. an explanation or justification) of what you did on a research project and why.

Intellectual Property
Honor patents, copyrights, and other forms of intellectual property. Do not use unpublished data, methods, or results without permission. Give proper acknowledgement or credit for all contributions to research. Never plagiarize.

Confidentiality
Protect confidential communications, such as papers or grants submitted for publication, personnel records, trade or military secrets, and patient records.

Responsible Publication
Publish in order to advance research and scholarship, not to advance just your own career. Avoid wasteful and duplicative publication.

Responsible Mentoring
Help to educate, mentor, and advise students. Promote their welfare and allow them to make their own decisions.

Respect for Colleagues
Respect your colleagues and treat them fairly.

Social Responsibility
Strive to promote social good and prevent or mitigate social harms through research, public education, and advocacy.

Non-Discrimination
Avoid discrimination against colleagues or students on the basis of sex, race, ethnicity, or other factors not related to scientific competence and integrity.

Maintain and improve your own professional competence and expertise through lifelong education and learning; take steps to promote competence in science as a whole.

Know and obey relevant laws and institutional and governmental policies.

Animal Care
Show proper respect and care for animals when using them in research. Do not conduct unnecessary or poorly designed animal experiments.

Human Subjects protection
When conducting research on human subjects, minimize harms and risks and maximize benefits; respect human dignity, privacy, and autonomy; take special precautions with vulnerable populations; and strive to distribute the benefits and burdens of research fairly.
* Adapted from Shamoo A and Resnik D. 2015. Responsible Conduct of Research, 3rd ed. (New York: Oxford University Press).
Ethical Decision Making in Research
Although codes, policies, and principles are very important and useful, like any set of rules, they do not cover every situation, they often conflict, and they require interpretation. It is therefore important for researchers to learn how to interpret, assess, and apply various research rules and how to make decisions and act ethically in various situations. The vast majority of decisions involve the straightforward application of ethical rules. For example, consider the following case:
The research protocol for a study of a drug on hypertension requires the administration of the drug at different doses to 50 laboratory mice, with chemical and behavioral tests to determine toxic effects. Tom has almost finished the experiment for Dr. Q. He has only 5 mice left to test. However, he really wants to finish his work in time to go to Florida on spring break with his friends, who are leaving tonight. He has injected the drug in all 50 mice but has not completed all of the tests. He therefore decides to extrapolate from the 45 completed results to produce the 5 additional results.
Many different research ethics policies would hold that Tom has acted unethically by fabricating data. If this study were sponsored by a federal agency, such as the NIH, his actions would constitute a form of research misconduct , which the government defines as "fabrication, falsification, or plagiarism" (or FFP). Actions that nearly all researchers classify as unethical are viewed as misconduct. It is important to remember, however, that misconduct occurs only when researchers intend to deceive : honest errors related to sloppiness, poor record keeping, miscalculations, bias, self-deception, and even negligence do not constitute misconduct. Also, reasonable disagreements about research methods, procedures, and interpretations do not constitute research misconduct. Consider the following case:
Dr. T has just discovered a mathematical error in his paper that has been accepted for publication in a journal. The error does not affect the overall results of his research, but it is potentially misleading. The journal has just gone to press, so it is too late to catch the error before it appears in print. In order to avoid embarrassment, Dr. T decides to ignore the error.
Dr. T's error is not misconduct nor is his decision to take no action to correct the error. Most researchers, as well as many different policies and codes would say that Dr. T should tell the journal (and any coauthors) about the error and consider publishing a correction or errata. Failing to publish a correction would be unethical because it would violate norms relating to honesty and objectivity in research.
There are many other activities that the government does not define as "misconduct" but which are still regarded by most researchers as unethical. These are sometimes referred to as " other deviations " from acceptable research practices and include:
- Publishing the same paper in two different journals without telling the editors
- Submitting the same paper to different journals without telling the editors
- Not informing a collaborator of your intent to file a patent in order to make sure that you are the sole inventor
- Including a colleague as an author on a paper in return for a favor even though the colleague did not make a serious contribution to the paper
- Discussing with your colleagues confidential data from a paper that you are reviewing for a journal
- Using data, ideas, or methods you learn about while reviewing a grant or a papers without permission
- Trimming outliers from a data set without discussing your reasons in paper
- Using an inappropriate statistical technique in order to enhance the significance of your research
- Bypassing the peer review process and announcing your results through a press conference without giving peers adequate information to review your work
- Conducting a review of the literature that fails to acknowledge the contributions of other people in the field or relevant prior work
- Stretching the truth on a grant application in order to convince reviewers that your project will make a significant contribution to the field
- Stretching the truth on a job application or curriculum vita
- Giving the same research project to two graduate students in order to see who can do it the fastest
- Overworking, neglecting, or exploiting graduate or post-doctoral students
- Failing to keep good research records
- Failing to maintain research data for a reasonable period of time
- Making derogatory comments and personal attacks in your review of author's submission
- Promising a student a better grade for sexual favors
- Using a racist epithet in the laboratory
- Making significant deviations from the research protocol approved by your institution's Animal Care and Use Committee or Institutional Review Board for Human Subjects Research without telling the committee or the board
- Not reporting an adverse event in a human research experiment
- Wasting animals in research
- Exposing students and staff to biological risks in violation of your institution's biosafety rules
- Sabotaging someone's work
- Stealing supplies, books, or data
- Rigging an experiment so you know how it will turn out
- Making unauthorized copies of data, papers, or computer programs
- Owning over $10,000 in stock in a company that sponsors your research and not disclosing this financial interest
- Deliberately overestimating the clinical significance of a new drug in order to obtain economic benefits
These actions would be regarded as unethical by most scientists and some might even be illegal in some cases. Most of these would also violate different professional ethics codes or institutional policies. However, they do not fall into the narrow category of actions that the government classifies as research misconduct. Indeed, there has been considerable debate about the definition of "research misconduct" and many researchers and policy makers are not satisfied with the government's narrow definition that focuses on FFP. However, given the huge list of potential offenses that might fall into the category "other serious deviations," and the practical problems with defining and policing these other deviations, it is understandable why government officials have chosen to limit their focus.
Finally, situations frequently arise in research in which different people disagree about the proper course of action and there is no broad consensus about what should be done. In these situations, there may be good arguments on both sides of the issue and different ethical principles may conflict. These situations create difficult decisions for research known as ethical or moral dilemmas . Consider the following case:
Dr. Wexford is the principal investigator of a large, epidemiological study on the health of 10,000 agricultural workers. She has an impressive dataset that includes information on demographics, environmental exposures, diet, genetics, and various disease outcomes such as cancer, Parkinson’s disease (PD), and ALS. She has just published a paper on the relationship between pesticide exposure and PD in a prestigious journal. She is planning to publish many other papers from her dataset. She receives a request from another research team that wants access to her complete dataset. They are interested in examining the relationship between pesticide exposures and skin cancer. Dr. Wexford was planning to conduct a study on this topic.
Dr. Wexford faces a difficult choice. On the one hand, the ethical norm of openness obliges her to share data with the other research team. Her funding agency may also have rules that obligate her to share data. On the other hand, if she shares data with the other team, they may publish results that she was planning to publish, thus depriving her (and her team) of recognition and priority. It seems that there are good arguments on both sides of this issue and Dr. Wexford needs to take some time to think about what she should do. One possible option is to share data, provided that the investigators sign a data use agreement. The agreement could define allowable uses of the data, publication plans, authorship, etc. Another option would be to offer to collaborate with the researchers.
The following are some step that researchers, such as Dr. Wexford, can take to deal with ethical dilemmas in research:
What is the problem or issue?
It is always important to get a clear statement of the problem. In this case, the issue is whether to share information with the other research team.
What is the relevant information?
Many bad decisions are made as a result of poor information. To know what to do, Dr. Wexford needs to have more information concerning such matters as university or funding agency or journal policies that may apply to this situation, the team's intellectual property interests, the possibility of negotiating some kind of agreement with the other team, whether the other team also has some information it is willing to share, the impact of the potential publications, etc.
What are the different options?
People may fail to see different options due to a limited imagination, bias, ignorance, or fear. In this case, there may be other choices besides 'share' or 'don't share,' such as 'negotiate an agreement' or 'offer to collaborate with the researchers.'
How do ethical codes or policies as well as legal rules apply to these different options?
The university or funding agency may have policies on data management that apply to this case. Broader ethical rules, such as openness and respect for credit and intellectual property, may also apply to this case. Laws relating to intellectual property may be relevant.
Are there any people who can offer ethical advice?
It may be useful to seek advice from a colleague, a senior researcher, your department chair, an ethics or compliance officer, or anyone else you can trust. In the case, Dr. Wexford might want to talk to her supervisor and research team before making a decision.
After considering these questions, a person facing an ethical dilemma may decide to ask more questions, gather more information, explore different options, or consider other ethical rules. However, at some point he or she will have to make a decision and then take action. Ideally, a person who makes a decision in an ethical dilemma should be able to justify his or her decision to himself or herself, as well as colleagues, administrators, and other people who might be affected by the decision. He or she should be able to articulate reasons for his or her conduct and should consider the following questions in order to explain how he or she arrived at his or her decision:
- Which choice will probably have the best overall consequences for science and society?
- Which choice could stand up to further publicity and scrutiny?
- Which choice could you not live with?
- Think of the wisest person you know. What would he or she do in this situation?
- Which choice would be the most just, fair, or responsible?
After considering all of these questions, one still might find it difficult to decide what to do. If this is the case, then it may be appropriate to consider others ways of making the decision, such as going with a gut feeling or intuition, seeking guidance through prayer or meditation, or even flipping a coin. Endorsing these methods in this context need not imply that ethical decisions are irrational, however. The main point is that human reasoning plays a pivotal role in ethical decision-making but there are limits to its ability to solve all ethical dilemmas in a finite amount of time.
Promoting Ethical Conduct in Science

Do U.S. research institutions meet or exceed federal mandates for instruction in responsible conduct of research? A national survey

Read about U.S. research instutuins follow federal manadates for ethics in research
Learn more about NIEHS Research
Most academic institutions in the US require undergraduate, graduate, or postgraduate students to have some education in the responsible conduct of research (RCR) . The NIH and NSF have both mandated training in research ethics for students and trainees. Many academic institutions outside of the US have also developed educational curricula in research ethics
Those of you who are taking or have taken courses in research ethics may be wondering why you are required to have education in research ethics. You may believe that you are highly ethical and know the difference between right and wrong. You would never fabricate or falsify data or plagiarize. Indeed, you also may believe that most of your colleagues are highly ethical and that there is no ethics problem in research..
If you feel this way, relax. No one is accusing you of acting unethically. Indeed, the evidence produced so far shows that misconduct is a very rare occurrence in research, although there is considerable variation among various estimates. The rate of misconduct has been estimated to be as low as 0.01% of researchers per year (based on confirmed cases of misconduct in federally funded research) to as high as 1% of researchers per year (based on self-reports of misconduct on anonymous surveys). See Shamoo and Resnik (2015), cited above.
Clearly, it would be useful to have more data on this topic, but so far there is no evidence that science has become ethically corrupt, despite some highly publicized scandals. Even if misconduct is only a rare occurrence, it can still have a tremendous impact on science and society because it can compromise the integrity of research, erode the public’s trust in science, and waste time and resources. Will education in research ethics help reduce the rate of misconduct in science? It is too early to tell. The answer to this question depends, in part, on how one understands the causes of misconduct. There are two main theories about why researchers commit misconduct. According to the "bad apple" theory, most scientists are highly ethical. Only researchers who are morally corrupt, economically desperate, or psychologically disturbed commit misconduct. Moreover, only a fool would commit misconduct because science's peer review system and self-correcting mechanisms will eventually catch those who try to cheat the system. In any case, a course in research ethics will have little impact on "bad apples," one might argue.
According to the "stressful" or "imperfect" environment theory, misconduct occurs because various institutional pressures, incentives, and constraints encourage people to commit misconduct, such as pressures to publish or obtain grants or contracts, career ambitions, the pursuit of profit or fame, poor supervision of students and trainees, and poor oversight of researchers (see Shamoo and Resnik 2015). Moreover, defenders of the stressful environment theory point out that science's peer review system is far from perfect and that it is relatively easy to cheat the system. Erroneous or fraudulent research often enters the public record without being detected for years. Misconduct probably results from environmental and individual causes, i.e. when people who are morally weak, ignorant, or insensitive are placed in stressful or imperfect environments. In any case, a course in research ethics can be useful in helping to prevent deviations from norms even if it does not prevent misconduct. Education in research ethics is can help people get a better understanding of ethical standards, policies, and issues and improve ethical judgment and decision making. Many of the deviations that occur in research may occur because researchers simply do not know or have never thought seriously about some of the ethical norms of research. For example, some unethical authorship practices probably reflect traditions and practices that have not been questioned seriously until recently. If the director of a lab is named as an author on every paper that comes from his lab, even if he does not make a significant contribution, what could be wrong with that? That's just the way it's done, one might argue. Another example where there may be some ignorance or mistaken traditions is conflicts of interest in research. A researcher may think that a "normal" or "traditional" financial relationship, such as accepting stock or a consulting fee from a drug company that sponsors her research, raises no serious ethical issues. Or perhaps a university administrator sees no ethical problem in taking a large gift with strings attached from a pharmaceutical company. Maybe a physician thinks that it is perfectly appropriate to receive a $300 finder’s fee for referring patients into a clinical trial.
If "deviations" from ethical conduct occur in research as a result of ignorance or a failure to reflect critically on problematic traditions, then a course in research ethics may help reduce the rate of serious deviations by improving the researcher's understanding of ethics and by sensitizing him or her to the issues.
Finally, education in research ethics should be able to help researchers grapple with the ethical dilemmas they are likely to encounter by introducing them to important concepts, tools, principles, and methods that can be useful in resolving these dilemmas. Scientists must deal with a number of different controversial topics, such as human embryonic stem cell research, cloning, genetic engineering, and research involving animal or human subjects, which require ethical reflection and deliberation.
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
Methodology
- Ethical Considerations in Research | Types & Examples
Ethical Considerations in Research | Types & Examples
Published on October 18, 2021 by Pritha Bhandari . Revised on May 9, 2024.
Ethical considerations in research are a set of principles that guide your research designs and practices. Scientists and researchers must always adhere to a certain code of conduct when collecting data from people.
The goals of human research often include understanding real-life phenomena, studying effective treatments, investigating behaviors, and improving lives in other ways. What you decide to research and how you conduct that research involve key ethical considerations.
These considerations work to
- protect the rights of research participants
- enhance research validity
- maintain scientific or academic integrity
Table of contents
Why do research ethics matter, getting ethical approval for your study, types of ethical issues, voluntary participation, informed consent, confidentiality, potential for harm, results communication, examples of ethical failures, other interesting articles, frequently asked questions about research ethics.
Research ethics matter for scientific integrity, human rights and dignity, and collaboration between science and society. These principles make sure that participation in studies is voluntary, informed, and safe for research subjects.
You’ll balance pursuing important research objectives with using ethical research methods and procedures. It’s always necessary to prevent permanent or excessive harm to participants, whether inadvertent or not.
Defying research ethics will also lower the credibility of your research because it’s hard for others to trust your data if your methods are morally questionable.
Even if a research idea is valuable to society, it doesn’t justify violating the human rights or dignity of your study participants.
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
Before you start any study involving data collection with people, you’ll submit your research proposal to an institutional review board (IRB) .
An IRB is a committee that checks whether your research aims and research design are ethically acceptable and follow your institution’s code of conduct. They check that your research materials and procedures are up to code.
If successful, you’ll receive IRB approval, and you can begin collecting data according to the approved procedures. If you want to make any changes to your procedures or materials, you’ll need to submit a modification application to the IRB for approval.
If unsuccessful, you may be asked to re-submit with modifications or your research proposal may receive a rejection. To get IRB approval, it’s important to explicitly note how you’ll tackle each of the ethical issues that may arise in your study.
There are several ethical issues you should always pay attention to in your research design, and these issues can overlap with each other.
You’ll usually outline ways you’ll deal with each issue in your research proposal if you plan to collect data from participants.
| Voluntary participation | Your participants are free to opt in or out of the study at any point in time. |
|---|---|
| Informed consent | Participants know the purpose, benefits, risks, and funding behind the study before they agree or decline to join. |
| Anonymity | You don’t know the identities of the participants. Personally identifiable data is not collected. |
| Confidentiality | You know who the participants are but you keep that information hidden from everyone else. You anonymize personally identifiable data so that it can’t be linked to other data by anyone else. |
| Potential for harm | Physical, social, psychological and all other types of harm are kept to an absolute minimum. |
| Results communication | You ensure your work is free of or research misconduct, and you accurately represent your results. |
Voluntary participation means that all research subjects are free to choose to participate without any pressure or coercion.
All participants are able to withdraw from, or leave, the study at any point without feeling an obligation to continue. Your participants don’t need to provide a reason for leaving the study.
It’s important to make it clear to participants that there are no negative consequences or repercussions to their refusal to participate. After all, they’re taking the time to help you in the research process , so you should respect their decisions without trying to change their minds.
Voluntary participation is an ethical principle protected by international law and many scientific codes of conduct.
Take special care to ensure there’s no pressure on participants when you’re working with vulnerable groups of people who may find it hard to stop the study even when they want to.
Informed consent refers to a situation in which all potential participants receive and understand all the information they need to decide whether they want to participate. This includes information about the study’s benefits, risks, funding, and institutional approval.
You make sure to provide all potential participants with all the relevant information about
- what the study is about
- the risks and benefits of taking part
- how long the study will take
- your supervisor’s contact information and the institution’s approval number
Usually, you’ll provide participants with a text for them to read and ask them if they have any questions. If they agree to participate, they can sign or initial the consent form. Note that this may not be sufficient for informed consent when you work with particularly vulnerable groups of people.
If you’re collecting data from people with low literacy, make sure to verbally explain the consent form to them before they agree to participate.
For participants with very limited English proficiency, you should always translate the study materials or work with an interpreter so they have all the information in their first language.
In research with children, you’ll often need informed permission for their participation from their parents or guardians. Although children cannot give informed consent, it’s best to also ask for their assent (agreement) to participate, depending on their age and maturity level.
Anonymity means that you don’t know who the participants are and you can’t link any individual participant to their data.
You can only guarantee anonymity by not collecting any personally identifying information—for example, names, phone numbers, email addresses, IP addresses, physical characteristics, photos, and videos.
In many cases, it may be impossible to truly anonymize data collection . For example, data collected in person or by phone cannot be considered fully anonymous because some personal identifiers (demographic information or phone numbers) are impossible to hide.
You’ll also need to collect some identifying information if you give your participants the option to withdraw their data at a later stage.
Data pseudonymization is an alternative method where you replace identifying information about participants with pseudonymous, or fake, identifiers. The data can still be linked to participants but it’s harder to do so because you separate personal information from the study data.
Confidentiality means that you know who the participants are, but you remove all identifying information from your report.
All participants have a right to privacy, so you should protect their personal data for as long as you store or use it. Even when you can’t collect data anonymously, you should secure confidentiality whenever you can.
Some research designs aren’t conducive to confidentiality, but it’s important to make all attempts and inform participants of the risks involved.
As a researcher, you have to consider all possible sources of harm to participants. Harm can come in many different forms.
- Psychological harm: Sensitive questions or tasks may trigger negative emotions such as shame or anxiety.
- Social harm: Participation can involve social risks, public embarrassment, or stigma.
- Physical harm: Pain or injury can result from the study procedures.
- Legal harm: Reporting sensitive data could lead to legal risks or a breach of privacy.
It’s best to consider every possible source of harm in your study as well as concrete ways to mitigate them. Involve your supervisor to discuss steps for harm reduction.
Make sure to disclose all possible risks of harm to participants before the study to get informed consent. If there is a risk of harm, prepare to provide participants with resources or counseling or medical services if needed.
Some of these questions may bring up negative emotions, so you inform participants about the sensitive nature of the survey and assure them that their responses will be confidential.
The way you communicate your research results can sometimes involve ethical issues. Good science communication is honest, reliable, and credible. It’s best to make your results as transparent as possible.
Take steps to actively avoid plagiarism and research misconduct wherever possible.
Plagiarism means submitting others’ works as your own. Although it can be unintentional, copying someone else’s work without proper credit amounts to stealing. It’s an ethical problem in research communication because you may benefit by harming other researchers.
Self-plagiarism is when you republish or re-submit parts of your own papers or reports without properly citing your original work.
This is problematic because you may benefit from presenting your ideas as new and original even though they’ve already been published elsewhere in the past. You may also be infringing on your previous publisher’s copyright, violating an ethical code, or wasting time and resources by doing so.
In extreme cases of self-plagiarism, entire datasets or papers are sometimes duplicated. These are major ethical violations because they can skew research findings if taken as original data.
You notice that two published studies have similar characteristics even though they are from different years. Their sample sizes, locations, treatments, and results are highly similar, and the studies share one author in common.
Research misconduct
Research misconduct means making up or falsifying data, manipulating data analyses, or misrepresenting results in research reports. It’s a form of academic fraud.
These actions are committed intentionally and can have serious consequences; research misconduct is not a simple mistake or a point of disagreement about data analyses.
Research misconduct is a serious ethical issue because it can undermine academic integrity and institutional credibility. It leads to a waste of funding and resources that could have been used for alternative research.
Later investigations revealed that they fabricated and manipulated their data to show a nonexistent link between vaccines and autism. Wakefield also neglected to disclose important conflicts of interest, and his medical license was taken away.
This fraudulent work sparked vaccine hesitancy among parents and caregivers. The rate of MMR vaccinations in children fell sharply, and measles outbreaks became more common due to a lack of herd immunity.
Research scandals with ethical failures are littered throughout history, but some took place not that long ago.
Some scientists in positions of power have historically mistreated or even abused research participants to investigate research problems at any cost. These participants were prisoners, under their care, or otherwise trusted them to treat them with dignity.
To demonstrate the importance of research ethics, we’ll briefly review two research studies that violated human rights in modern history.
These experiments were inhumane and resulted in trauma, permanent disabilities, or death in many cases.
After some Nazi doctors were put on trial for their crimes, the Nuremberg Code of research ethics for human experimentation was developed in 1947 to establish a new standard for human experimentation in medical research.
In reality, the actual goal was to study the effects of the disease when left untreated, and the researchers never informed participants about their diagnoses or the research aims.
Although participants experienced severe health problems, including blindness and other complications, the researchers only pretended to provide medical care.
When treatment became possible in 1943, 11 years after the study began, none of the participants were offered it, despite their health conditions and high risk of death.
Ethical failures like these resulted in severe harm to participants, wasted resources, and lower trust in science and scientists. This is why all research institutions have strict ethical guidelines for performing research.
If you want to know more about statistics , methodology , or research bias , make sure to check out some of our other articles with explanations and examples.
- Normal distribution
- Measures of central tendency
- Chi square tests
- Confidence interval
- Quartiles & Quantiles
- Cluster sampling
- Stratified sampling
- Thematic analysis
- Cohort study
- Peer review
- Ethnography
Research bias
- Implicit bias
- Cognitive bias
- Conformity bias
- Hawthorne effect
- Availability heuristic
- Attrition bias
- Social desirability bias
Ethical considerations in research are a set of principles that guide your research designs and practices. These principles include voluntary participation, informed consent, anonymity, confidentiality, potential for harm, and results communication.
Scientists and researchers must always adhere to a certain code of conduct when collecting data from others .
These considerations protect the rights of research participants, enhance research validity , and maintain scientific integrity.
Research ethics matter for scientific integrity, human rights and dignity, and collaboration between science and society. These principles make sure that participation in studies is voluntary, informed, and safe.
Anonymity means you don’t know who the participants are, while confidentiality means you know who they are but remove identifying information from your research report. Both are important ethical considerations .
You can only guarantee anonymity by not collecting any personally identifying information—for example, names, phone numbers, email addresses, IP addresses, physical characteristics, photos, or videos.
You can keep data confidential by using aggregate information in your research report, so that you only refer to groups of participants rather than individuals.
These actions are committed intentionally and can have serious consequences; research misconduct is not a simple mistake or a point of disagreement but a serious ethical failure.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
Bhandari, P. (2024, May 09). Ethical Considerations in Research | Types & Examples. Scribbr. Retrieved September 13, 2024, from https://www.scribbr.com/methodology/research-ethics/
Is this article helpful?

Pritha Bhandari
Other students also liked, data collection | definition, methods & examples, what is self-plagiarism | definition & how to avoid it, how to avoid plagiarism | tips on citing sources, get unlimited documents corrected.
✔ Free APA citation check included ✔ Unlimited document corrections ✔ Specialized in correcting academic texts

Site Search
- How to Search
- Advisory Group
- Editorial Board
- OEC Fellows
- History and Funding
- Using OEC Materials
- Collections
- Research Ethics Resources
- Ethics Projects
- Communities of Practice
- Get Involved
- Submit Content
- Open Access Membership
- Become a Partner
- Advanced Search
- Webinar Series
Introduction: What is Research Ethics?
Research Ethics is defined here to be the ethics of the planning, conduct, and reporting of research. This introduction covers what research ethics is, its ethical distinctions, approaches to teaching research ethics, and other resources on this topic.
What is Research Ethics
Why Teach Research Ethics
Animal Subjects
Biosecurity
Collaboration
Conflicts of Interest
Data Management
Human Subjects
Peer Review
Publication
Research Misconduct
Social Responsibility
Stem Cell Research
Whistleblowing
Descriptions of educational settings , including in the classroom, and in research contexts.
Case Studies
Other Discussion Tools
Information about the history and authors of the Resources for Research Ethics Collection
What is Research Ethics?
Research Ethics is defined here to be the ethics of the planning, conduct, and reporting of research. It is clear that research ethics should include:
- Protections of human and animal subjects
However, not all researchers use human or animal subjects, nor are the ethical dimensions of research confined solely to protections for research subjects. Other ethical challenges are rooted in many dimensions of research, including the:
- Collection, use, and interpretation of research data
- Methods for reporting and reviewing research plans or findings
- Relationships among researchers with one another
- Relationships between researchers and those that will be affected by their research
- Means for responding to misunderstandings, disputes, or misconduct
- Options for promoting ethical conduct in research
The domain of research ethics is intended to include nothing less than the fostering of research that protects the interests of the public, the subjects of research, and the researchers themselves.
Ethical Distinctions
In discussing or teaching research ethics, it is important to keep some basic distinctions in mind.
- It is important not to confuse moral claims about how people ought to behave with descriptive claims about how they in fact do behave. From the fact that gift authorship or signing off on un-reviewed data may be "common practice" in some contexts, it doesn't follow that they are morally or professionally justified. Nor is morality to be confused with the moral beliefs or ethical codes that a given group or society holds (how some group thinks people should live). A belief in segregation is not morally justified simply because it is widely held by a group of people or given society. Philosophers term this distinction between prescriptive and descriptive claims the 'is-ought distinction.'
- A second important distinction is that between morality and the law. The law may or may not conform to the demands of ethics (Kagan, 1998). To take a contemporary example: many believe that the law prohibiting federally funded stem cell research is objectionable on moral (as well as scientific) grounds, i.e., that such research can save lives and prevent much human misery. History is full of examples of bad laws, that is laws now regarded as morally unjustifiable, e.g., the laws of apartheid, laws prohibiting women from voting or inter-racial couples from marrying.
- It is also helpful to distinguish between two different levels of discussion (or two different kinds of ethical questions): first-order or "ground-level" questions and second-order questions.
- First-order moral questions concern what we should do. Such questions may be very general or quite specific. One might ask whether the tradition of 'senior' authorship should be defended and preserved or, more generally, what are the principles that should go into deciding the issue of 'senior' authorship. Such questions and the substantive proposals regarding how to answer them belong to the domain of what moral philosophers call 'normative ethics.'
- Second-order moral questions concern the nature and purpose of morality itself. When someone claims that falsifying data is wrong, what exactly is the standing of this claim? What exactly does the word 'wrong' mean in the conduct of scientific research? And what are we doing when we make claims about right and wrong, scientific integrity and research misconduct? These second-order questions are quite different from the ground-level questions about how to conduct one's private or professional life raised above. They concern the nature of morality rather than its content, i.e., what acts are required, permitted or prohibited. This is the domain of what moral philosophers call 'metaethics' (Kagan, 1998).
Ethical Approaches
Each of these approaches provides moral principles and ways of thinking about the responsibilities, duties and obligations of moral life. Individually and jointly, they can provide practical guidance in ethical decision-making.
- One of the most influential and familiar approaches to ethics is deontological ethics, associated with Immanuel Kant (1742-1804). Deontological ethics hold certain acts as right or wrong in themselves, e.g., promise breaking or lying. So, for example, in the context of research, fraud, plagiarism and misrepresentation are regarded as morally wrong in themselves, not simply because they (tend to) have bad consequences. The deontological approach is generally grounded in a single fundamental principle: Act as you would wish others to act towards you OR always treat persons as an end, never as a means to an end.
- From such central principles are derived rules or guidelines for what is permitted, required and prohibited. Objections to principle-based or deontological ethics include the difficulty of applying highly general principles to specific cases, e.g.: Does treating persons as ends rule out physician-assisted suicide, or require it? Deontological ethics is generally contrasted to consequentialist ethics (Honderich, 1995).
- According to consequentialist approaches, the rightness or wrongness of an action depends solely on its consequences. One should act in such a way as to bring about the best state of affairs, where the best state of affairs may be understood in various ways, e.g., as the greatest happiness for the greatest number of people, maximizing pleasure and minimizing pain or maximizing the satisfaction of preferences. A theory such as Utilitarianism (with its roots in the work of Jeremy Bentham and John Stuart Mill) is generally taken as the paradigm example of consequentialism. Objections to consequentialist ethics tend to focus on its willingness to regard individual rights and values as "negotiable." So, for example, most people would regard murder as wrong independently of the fact that killing one person might allow several others to be saved (the infamous sacrifice of an ailing patient to provide organs for several other needy patients). Similarly, widespread moral opinion holds certain values important (integrity, justice) not only because they generally lead to good outcomes, but in and of themselves.
- Virtue ethics focuses on moral character rather than action and behavior considered in isolation. Central to this approach is the question what ought we (as individuals, as scientists, as physicians) to be rather than simply what we ought to do. The emphasis here is on inner states, that is, moral dispositions and habits such as courage or a developed sense of personal integrity. Virtue ethics can be a useful approach in the context of RCR and professional ethics, emphasizing the importance of moral virtues such as compassion, honesty, and respect. This approach has also a great deal to offer in discussions of bioethical issues where a traditional emphasis on rights and abstract principles frequently results in polarized, stalled discussions (e.g., abortion debates contrasting the rights of the mother against the rights of the fetus).
- The term 'an ethics of care' grows out of the work of Carol Gilligan, whose empirical work in moral psychology claimed to discover a "different voice," a mode of moral thinking distinct from principle-based moral thinking (e.g., the theories of Kant and Mill). An ethics of care stresses compassion and empathetic understanding, virtues Gilligan associated with traditional care-giving roles, especially those of women.
- This approach differs from traditional moral theories in two important ways. First, it assumes that it is the connections between persons, e.g., lab teams, colleagues, parents and children, student and mentor, not merely the rights and obligations of discrete individuals that matter. The moral world, on this view, is best seen not as the interaction of discrete individuals, each with his or her own interests and rights, but as an interrelated web of obligations and commitment. We interact, much of the time, not as private individuals, but as members of families, couples, institutions, research groups, a given profession and so on. Second, these human relationships, including relationships of dependency, play a crucial role on this account in determining what our moral obligations and responsibilities are. So, for example, individuals have special responsibilities to care for their children, students, patients, and research subjects.
- An ethics of care is thus particularly useful in discussing human and animal subjects research, issues of informed consent, and the treatment of vulnerable populations such as children, the infirm or the ill.
- The case study approach begins from real or hypothetical cases. Its objective is to identify the intuitively plausible principles that should be taken into account in resolving the issues at hand. The case study approach then proceeds to critically evaluate those principles. In discussing whistle-blowing, for example, a good starting point is with recent cases of research misconduct, seeking to identify and evaluate principles such as a commitment to the integrity of science, protecting privacy, or avoiding false or unsubstantiated charges. In the context of RCR instruction, case studies provide one of the most interesting and effective approaches to developing sensitivity to ethical issues and to honing ethical decision-making skills.
- Strictly speaking, casuistry is more properly understood as a method for doing ethics rather than as itself an ethical theory. However, casuistry is not wholly unconnected to ethical theory. The need for a basis upon which to evaluate competing principles, e.g., the importance of the well-being of an individual patient vs. a concern for just allocation of scarce medical resources, makes ethical theory relevant even with case study approaches.
- Applied ethics is a branch of normative ethics. It deals with practical questions particularly in relation to the professions. Perhaps the best known area of applied ethics is bioethics, which deals with ethical questions arising in medicine and the biological sciences, e.g., questions concerning the application of new areas of technology (stem cells, cloning, genetic screening, nanotechnology, etc.), end of life issues, organ transplants, and just distribution of healthcare. Training in responsible conduct of research or "research ethics" is merely one among various forms of professional ethics that have come to prominence since the 1960s. Worth noting, however, is that concern with professional ethics is not new, as ancient codes such as the Hippocratic Oath and guild standards attest (Singer, 1986).
- Adams D, Pimple KD (2005): Research Misconduct and Crime: Lessons from Criminal Science on Preventing Misconduct and Promoting Integrity. Accountability in Research 12(3):225-240.
- Anderson MS, Horn AS, Risbey KR, Ronning EA, De Vries R, Martinson BC (2007): What Do Mentoring and Training in the Responsible Conduct of Research Have To Do with Scientists' Misbehavior? Findings from a National Survey of NIH-Funded Scientists . Academic Medicine 82(9):853-860.
- Bulger RE, Heitman E (2007): Expanding Responsible Conduct of Research Instruction across the University. Academic Medicine. 82(9):876-878.
- Kalichman MW (2006): Ethics and Science: A 0.1% solution. Issues in Science and Technology 23:34-36.
- Kalichman MW (2007): Responding to Challenges in Educating for the Responsible Conduct of Research, Academic Medicine. 82(9):870-875.
- Kalichman MW, Plemmons DK (2007): Reported Goals for Responsible Conduct of Research Courses. Academic Medicine. 82(9):846-852.
- Kalichman MW (2009): Evidence-based research ethics. The American Journal of Bioethics 9(6&7): 85-87.
- Pimple KD (2002): Six Domains of Research Ethics: A Heuristic Framework for the Responsible Conduct of Research. Science and Engineering Ethics 8(2):191-205.
- Steneck NH (2006): Fostering Integrity in Research: Definitions, Current Knowledge, and Future Directions. Science and Engineering Ethics 12:53-74.
- Steneck NH, Bulger RE (2007): The History, Purpose, and Future of Instruction in the Responsible Conduct of Research. Academic Medicine. 82(9):829-834.
- Vasgird DR (2007): Prevention over Cure: The Administrative Rationale for Education in the Responsible Conduct of Research. Academic Medicine. 82(9):835-837.
- Aristotle. The Nichomachean Ethics.
- Beauchamp RL, Childress JF (2001): Principles of Biomedical Ethics, 5th edition, NY: Oxford University Press.
- Bentham, J (1781): An Introduction to the Principles of Morals and Legislation.
- Gilligan C (1993): In a Different Voice: Psychological Theory and Women's Development. Cambridge: Harvard University Press.
- Glover, Jonathan (1977): Penguin Books.
- Honderich T, ed. (1995): The Oxford Companion to Philosophy, Oxford and New York: Oxford University Press.
- Kagan S (1998): Normative Ethics. Westview Press.
- Kant I (1785): Groundwork of the Metaphysics of Morals.
- Kant I (1788): Critique of Practical Reason.
- Kant I (1797): The Metaphysics of Morals.
- Kant I (1797): On a Supposed right to Lie from Benevolent Motives.
- Kuhse H, Singer P (1999): Bioethics: An Anthology. Blackwell Publishers.
- Mill JS (1861): Utilitarianism.
- Rachels J (1999): The Elements of Moral Philosophy, 3rd edition, Boston: McGraw-Hill.
- Regan T (1993): Matters of Life and Death: New Introductory Essays in Moral Philosophy, 3rd edition. New York: McGraw-Hill. The history of ethics.
- Singer P (1993): Practical Ethics, 2nd ed. Cambridge University Press.
The Resources for Research Ethics Education site was originally developed and maintained by Dr. Michael Kalichman, Director of the Research Ethics Program at the University of California San Diego. The site was transferred to the Online Ethics Center in 2021 with the permission of the author.
Related Resources
Submit Content to the OEC Donate

This material is based upon work supported by the National Science Foundation under Award No. 2055332. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation.

Research Ethics & Ethical Considerations
A Plain-Language Explainer With Examples
By: Derek Jansen (MBA) | Reviewers: Dr Eunice Rautenbach | May 2024
Research ethics are one of those “ unsexy but essential ” subjects that you need to fully understand (and apply) to conquer your dissertation, thesis or research paper. In this post, we’ll unpack research ethics using plain language and loads of examples .
Overview: Research Ethics 101
- What are research ethics?
- Why should you care?
- Research ethics principles
- Respect for persons
- Beneficence
- Objectivity
- Key takeaways
What (exactly) are research ethics?
At the simplest level, research ethics are a set of principles that ensure that your study is conducted responsibly, safely, and with integrity. More specifically, research ethics help protect the rights and welfare of your research participants, while also ensuring the credibility of your research findings.
Research ethics are critically important for a number of reasons:
Firstly, they’re a complete non-negotiable when it comes to getting your research proposal approved. Pretty much all universities will have a set of ethical criteria that student projects need to adhere to – and these are typically very strictly enforced. So, if your proposed study doesn’t tick the necessary ethical boxes, it won’t be approved .
Beyond the practical aspect of approval, research ethics are essential as they ensure that your study’s participants (whether human or animal) are properly protected . In turn, this fosters trust between you and your participants – as well as trust between researchers and the public more generally. As you can probably imagine, it wouldn’t be good if the general public had a negative perception of researchers!
Last but not least, research ethics help ensure that your study’s results are valid and reliable . In other words, that you measured the thing you intended to measure – and that other researchers can repeat your study. If you’re not familiar with the concepts of reliability and validity , we’ve got a straightforward explainer video covering that below.
The Core Principles
In practical terms, each university or institution will have its own ethics policy – so, what exactly constitutes “ethical research” will vary somewhat between institutions and countries. Nevertheless, there are a handful of core principles that shape ethics policies. These principles include:
Let’s unpack each of these to make them a little more tangible.
Ethics Principle 1: Respect for persons
As the name suggests, this principle is all about ensuring that your participants are treated fairly and respectfully . In practical terms, this means informed consent – in other words, participants should be fully informed about the nature of the research, as well as any potential risks. Additionally, they should be able to withdraw from the study at any time. This is especially important when you’re dealing with vulnerable populations – for example, children, the elderly or people with cognitive disabilities.
Another dimension of the “respect for persons” principle is confidentiality and data protection . In other words, your participants’ personal information should be kept strictly confidential and secure at all times. Depending on the specifics of your project, this might also involve anonymising or masking people’s identities. As mentioned earlier, the exact requirements will vary between universities, so be sure to thoroughly review your institution’s ethics policy before you start designing your project.
Need a helping hand?
Ethics Principle 2: Beneficence
This principle is a little more opaque, but in simple terms beneficence means that you, as the researcher, should aim to maximise the benefits of your work, while minimising any potential harm to your participants.
In practical terms, benefits could include advancing knowledge, improving health outcomes, or providing educational value. Conversely, potential harms could include:
- Physical harm from accidents or injuries
- Psychological harm, such as stress or embarrassment
- Social harm, such as stigmatisation or loss of reputation
- Economic harm – in other words, financial costs or lost income
Simply put, the beneficence principle means that researchers must always try to identify potential risks and take suitable measures to reduce or eliminate them.

Ethics Principle 3: Objectivity
As you can probably guess, this principle is all about attempting to minimise research bias to the greatest degree possible. In other words, you’ll need to reduce subjectivity and increase objectivity wherever possible.
In practical terms, this principle has the largest impact on the methodology of your study – specifically the data collection and data analysis aspects. For example, you’ll need to ensure that the selection of your participants (in other words, your sampling strategy ) is aligned with your research aims – and that your sample isn’t skewed in a way that supports your presuppositions.
If you’re keen to learn more about research bias and the various ways in which you could unintentionally skew your results, check out the video below.
Ethics Principle 4: Integrity
Again, no surprises here; this principle is all about producing “honest work” . It goes without saying that researchers should always conduct their work honestly and transparently, report their findings accurately, and disclose any potential conflicts of interest upfront.
This is all pretty obvious, but another aspect of the integrity principle that’s sometimes overlooked is respect for intellectual property . In practical terms, this means you need to honour any patents, copyrights, or other forms of intellectual property that you utilise while undertaking your research. Along the same vein, you shouldn’t use any unpublished data, methods, or results without explicit, written permission from the respective owner.
Linked to all of this is the broader issue of plagiarism . Needless to say, if you’re drawing on someone else’s published work, be sure to cite your sources, in the correct format. To make life easier, use a reference manager such as Mendeley or Zotero to ensure that your citations and reference list are perfectly polished.
FAQs: Research Ethics
Research ethics & ethical considertation, what is informed consent.
Informed consent simply means providing your potential participants with all necessary information about the study. This should include information regarding the study’s purpose, procedures, risks, and benefits. This information allows your potential participants to make a voluntary and informed decision about whether to participate.
How should I obtain consent from non-English speaking participants?
What about animals.
When conducting research with animals, ensure you adhere to ethical guidelines for the humane treatment of animals. Again, the exact requirements here will vary between institutions, but typically include minimising pain and distress, using alternatives where possible, and obtaining approval from an animal care and use committee.
What is the role of the ERB or IRB?
An ethics review board (ERB) or institutional review board (IRB) evaluates research proposals to ensure they meet ethical standards. The board reviews study designs, consent forms, and data handling procedures, to protect participants’ welfare and rights.
How can I obtain ethical approval for my project?
This varies between universities, but you will typically need to submit a detailed research proposal to your institution’s ethics committee. This proposal should include your research objectives, methods, and how you plan to address ethical considerations like informed consent, confidentiality, and risk minimisation. You can learn more about how to write a proposal here .
How do I ensure ethical collaboration when working with colleagues?
Collaborative research should be conducted with mutual respect and clear agreements on roles, contributions, and publication credits. Open communication is key to preventing conflicts and misunderstandings. Also, be sure to check whether your university has any specific requirements with regards to collaborative efforts and division of labour.
How should I address ethical concerns relating to my funding source?
Key takeaways: research ethics 101.
Here’s a quick recap of the key points we’ve covered:
- Research ethics are a set of principles that ensure that your study is conducted responsibly.
- It’s essential that you design your study around these principles, or it simply won’t get approved.
- The four ethics principles we looked at are: respect for persons, beneficence, objectivity and integrity
As mentioned, the exact requirements will vary slightly depending on the institution and country, so be sure to thoroughly review your university’s research ethics policy before you start developing your study.

Psst... there’s more!
This post was based on one of our popular Research Bootcamps . If you're working on a research project, you'll definitely want to check this out ...
Great piece!!!
Submit a Comment Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
- Print Friendly
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, automatically generate references for free.
- Knowledge Base
- Methodology
- Ethical Considerations in Research | Types & Examples
Ethical Considerations in Research | Types & Examples
Published on 7 May 2022 by Pritha Bhandari . Revised on 6 July 2024.
Ethical considerations in research are a set of principles that guide your research designs and practices. Scientists and researchers must always adhere to a certain code of conduct when collecting data from people.
The goals of human research often include understanding real-life phenomena, studying effective treatments, investigating behaviours, and improving lives in other ways. What you decide to research and how you conduct that research involve key ethical considerations.
These considerations work to:
- Protect the rights of research participants
- Enhance research validity
- Maintain scientific integrity
Table of contents
Why do research ethics matter, getting ethical approval for your study, types of ethical issues, voluntary participation, informed consent, confidentiality, potential for harm, results communication, examples of ethical failures, frequently asked questions about research ethics.
Research ethics matter for scientific integrity, human rights and dignity, and collaboration between science and society. These principles make sure that participation in studies is voluntary, informed, and safe for research subjects.
You’ll balance pursuing important research aims with using ethical research methods and procedures. It’s always necessary to prevent permanent or excessive harm to participants, whether inadvertent or not.
Defying research ethics will also lower the credibility of your research because it’s hard for others to trust your data if your methods are morally questionable.
Even if a research idea is valuable to society, it doesn’t justify violating the human rights or dignity of your study participants.
Prevent plagiarism, run a free check.
Before you start any study involving data collection with people, you’ll submit your research proposal to an institutional review board (IRB) .
An IRB is a committee that checks whether your research aims and research design are ethically acceptable and follow your institution’s code of conduct. They check that your research materials and procedures are up to code.
If successful, you’ll receive IRB approval, and you can begin collecting data according to the approved procedures. If you want to make any changes to your procedures or materials, you’ll need to submit a modification application to the IRB for approval.
If unsuccessful, you may be asked to re-submit with modifications or your research proposal may receive a rejection. To get IRB approval, it’s important to explicitly note how you’ll tackle each of the ethical issues that may arise in your study.
There are several ethical issues you should always pay attention to in your research design, and these issues can overlap with each other.
You’ll usually outline ways you’ll deal with each issue in your research proposal if you plan to collect data from participants.
| Voluntary participation | Your participants are free to opt in or out of the study at any point in time. |
|---|---|
| Informed consent | Participants know the purpose, benefits, risks, and funding behind the study before they agree or decline to join. |
| Anonymity | You don’t know the identities of the participants. Personally identifiable data is not collected. |
| Confidentiality | You know who the participants are but keep that information hidden from everyone else. You anonymise personally identifiable data so that it can’t be linked to other data by anyone else. |
| Potential for harm | Physical, social, psychological, and all other types of harm are kept to an absolute minimum. |
| Results communication | You ensure your work is free of plagiarism or research misconduct, and you accurately represent your results. |
Voluntary participation means that all research subjects are free to choose to participate without any pressure or coercion.
All participants are able to withdraw from, or leave, the study at any point without feeling an obligation to continue. Your participants don’t need to provide a reason for leaving the study.
It’s important to make it clear to participants that there are no negative consequences or repercussions to their refusal to participate. After all, they’re taking the time to help you in the research process, so you should respect their decisions without trying to change their minds.
Voluntary participation is an ethical principle protected by international law and many scientific codes of conduct.
Take special care to ensure there’s no pressure on participants when you’re working with vulnerable groups of people who may find it hard to stop the study even when they want to.
Informed consent refers to a situation in which all potential participants receive and understand all the information they need to decide whether they want to participate. This includes information about the study’s benefits, risks, funding, and institutional approval.
- What the study is about
- The risks and benefits of taking part
- How long the study will take
- Your supervisor’s contact information and the institution’s approval number
Usually, you’ll provide participants with a text for them to read and ask them if they have any questions. If they agree to participate, they can sign or initial the consent form. Note that this may not be sufficient for informed consent when you work with particularly vulnerable groups of people.
If you’re collecting data from people with low literacy, make sure to verbally explain the consent form to them before they agree to participate.
For participants with very limited English proficiency, you should always translate the study materials or work with an interpreter so they have all the information in their first language.
In research with children, you’ll often need informed permission for their participation from their parents or guardians. Although children cannot give informed consent, it’s best to also ask for their assent (agreement) to participate, depending on their age and maturity level.
Anonymity means that you don’t know who the participants are and you can’t link any individual participant to their data.
You can only guarantee anonymity by not collecting any personally identifying information – for example, names, phone numbers, email addresses, IP addresses, physical characteristics, photos, and videos.
In many cases, it may be impossible to truly anonymise data collection. For example, data collected in person or by phone cannot be considered fully anonymous because some personal identifiers (demographic information or phone numbers) are impossible to hide.
You’ll also need to collect some identifying information if you give your participants the option to withdraw their data at a later stage.
Data pseudonymisation is an alternative method where you replace identifying information about participants with pseudonymous, or fake, identifiers. The data can still be linked to participants, but it’s harder to do so because you separate personal information from the study data.
Confidentiality means that you know who the participants are, but you remove all identifying information from your report.
All participants have a right to privacy, so you should protect their personal data for as long as you store or use it. Even when you can’t collect data anonymously, you should secure confidentiality whenever you can.
Some research designs aren’t conducive to confidentiality, but it’s important to make all attempts and inform participants of the risks involved.
As a researcher, you have to consider all possible sources of harm to participants. Harm can come in many different forms.
- Psychological harm: Sensitive questions or tasks may trigger negative emotions such as shame or anxiety.
- Social harm: Participation can involve social risks, public embarrassment, or stigma.
- Physical harm: Pain or injury can result from the study procedures.
- Legal harm: Reporting sensitive data could lead to legal risks or a breach of privacy.
It’s best to consider every possible source of harm in your study, as well as concrete ways to mitigate them. Involve your supervisor to discuss steps for harm reduction.
Make sure to disclose all possible risks of harm to participants before the study to get informed consent. If there is a risk of harm, prepare to provide participants with resources, counselling, or medical services if needed.
Some of these questions may bring up negative emotions, so you inform participants about the sensitive nature of the survey and assure them that their responses will be confidential.
The way you communicate your research results can sometimes involve ethical issues. Good science communication is honest, reliable, and credible. It’s best to make your results as transparent as possible.
Take steps to actively avoid plagiarism and research misconduct wherever possible.
Plagiarism means submitting others’ works as your own. Although it can be unintentional, copying someone else’s work without proper credit amounts to stealing. It’s an ethical problem in research communication because you may benefit by harming other researchers.
Self-plagiarism is when you republish or re-submit parts of your own papers or reports without properly citing your original work.
This is problematic because you may benefit from presenting your ideas as new and original even though they’ve already been published elsewhere in the past. You may also be infringing on your previous publisher’s copyright, violating an ethical code, or wasting time and resources by doing so.
In extreme cases of self-plagiarism, entire datasets or papers are sometimes duplicated. These are major ethical violations because they can skew research findings if taken as original data.
You notice that two published studies have similar characteristics even though they are from different years. Their sample sizes, locations, treatments, and results are highly similar, and the studies share one author in common.
Research misconduct
Research misconduct means making up or falsifying data, manipulating data analyses, or misrepresenting results in research reports. It’s a form of academic fraud.
These actions are committed intentionally and can have serious consequences; research misconduct is not a simple mistake or a point of disagreement about data analyses.
Research misconduct is a serious ethical issue because it can undermine scientific integrity and institutional credibility. It leads to a waste of funding and resources that could have been used for alternative research.
Later investigations revealed that they fabricated and manipulated their data to show a nonexistent link between vaccines and autism. Wakefield also neglected to disclose important conflicts of interest, and his medical license was taken away.
This fraudulent work sparked vaccine hesitancy among parents and caregivers. The rate of MMR vaccinations in children fell sharply, and measles outbreaks became more common due to a lack of herd immunity.
Research scandals with ethical failures are littered throughout history, but some took place not that long ago.
Some scientists in positions of power have historically mistreated or even abused research participants to investigate research problems at any cost. These participants were prisoners, under their care, or otherwise trusted them to treat them with dignity.
To demonstrate the importance of research ethics, we’ll briefly review two research studies that violated human rights in modern history.
These experiments were inhumane and resulted in trauma, permanent disabilities, or death in many cases.
After some Nazi doctors were put on trial for their crimes, the Nuremberg Code of research ethics for human experimentation was developed in 1947 to establish a new standard for human experimentation in medical research.
In reality, the actual goal was to study the effects of the disease when left untreated, and the researchers never informed participants about their diagnoses or the research aims.
Although participants experienced severe health problems, including blindness and other complications, the researchers only pretended to provide medical care.
When treatment became possible in 1943, 11 years after the study began, none of the participants were offered it, despite their health conditions and high risk of death.
Ethical failures like these resulted in severe harm to participants, wasted resources, and lower trust in science and scientists. This is why all research institutions have strict ethical guidelines for performing research.
Ethical considerations in research are a set of principles that guide your research designs and practices. These principles include voluntary participation, informed consent, anonymity, confidentiality, potential for harm, and results communication.
Scientists and researchers must always adhere to a certain code of conduct when collecting data from others .
These considerations protect the rights of research participants, enhance research validity , and maintain scientific integrity.
Research ethics matter for scientific integrity, human rights and dignity, and collaboration between science and society. These principles make sure that participation in studies is voluntary, informed, and safe.
Anonymity means you don’t know who the participants are, while confidentiality means you know who they are but remove identifying information from your research report. Both are important ethical considerations .
You can only guarantee anonymity by not collecting any personally identifying information – for example, names, phone numbers, email addresses, IP addresses, physical characteristics, photos, or videos.
You can keep data confidential by using aggregate information in your research report, so that you only refer to groups of participants rather than individuals.
These actions are committed intentionally and can have serious consequences; research misconduct is not a simple mistake or a point of disagreement but a serious ethical failure.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the ‘Cite this Scribbr article’ button to automatically add the citation to our free Reference Generator.
Bhandari, P. (2024, July 05). Ethical Considerations in Research | Types & Examples. Scribbr. Retrieved 9 September 2024, from https://www.scribbr.co.uk/research-methods/ethical-considerations/
Is this article helpful?

Pritha Bhandari
Other students also liked, a quick guide to experimental design | 5 steps & examples, data collection methods | step-by-step guide & examples, how to avoid plagiarism | tips on citing sources.
- Original article
- Open access
- Published: 13 July 2021
Assisting you to advance with ethics in research: an introduction to ethical governance and application procedures
- Shivadas Sivasubramaniam 1 ,
- Dita Henek Dlabolová 2 ,
- Veronika Kralikova 3 &
- Zeenath Reza Khan 3
International Journal for Educational Integrity volume 17 , Article number: 14 ( 2021 ) Cite this article
20k Accesses
14 Citations
4 Altmetric
Metrics details
Ethics and ethical behaviour are the fundamental pillars of a civilised society. The focus on ethical behaviour is indispensable in certain fields such as medicine, finance, or law. In fact, ethics gets precedence with anything that would include, affect, transform, or influence upon individuals, communities or any living creatures. Many institutions within Europe have set up their own committees to focus on or approve activities that have ethical impact. In contrast, lesser-developed countries (worldwide) are trying to set up these committees to govern their academia and research. As the first European consortium established to assist academic integrity, European Network for Academic Integrity (ENAI), we felt the importance of guiding those institutions and communities that are trying to conduct research with ethical principles. We have established an ethical advisory working group within ENAI with the aim to promote ethics within curriculum, research and institutional policies. We are constantly researching available data on this subject and committed to help the academia to convey and conduct ethical behaviour. Upon preliminary review and discussion, the group found a disparity in understanding, practice and teaching approaches to ethical applications of research projects among peers. Therefore, this short paper preliminarily aims to critically review the available information on ethics, the history behind establishing ethical principles and its international guidelines to govern research.
The paper is based on the workshop conducted in the 5th International conference Plagiarism across Europe and Beyond, in Mykolas Romeris University, Lithuania in 2019. During the workshop, we have detailed a) basic needs of an ethical committee within an institution; b) a typical ethical approval process (with examples from three different universities); and c) the ways to obtain informed consent with some examples. These are summarised in this paper with some example comparisons of ethical approval processes from different universities. We believe this paper will provide guidelines on preparing and training both researchers and research students in appropriately upholding ethical practices through ethical approval processes.
Introduction
Ethics and ethical behaviour (often linked to “responsible practice”) are the fundamental pillars of a civilised society. Ethical behaviour with integrity is important to maintain academic and research activities. It affects everything we do, and gets precedence with anything that would include/affect, transform, or impact upon individuals, communities or any living creatures. In other words, ethics would help us improve our living standards (LaFollette, 2007 ). The focus on ethical behaviour is indispensable in certain fields such as medicine, finance, or law, but is also gaining recognition in all disciplines engaged in research. Therefore, institutions are expected to develop ethical guidelines in research to maintain quality, initiate/own integrity and above all be transparent to be successful by limiting any allegation of misconduct (Flite and Harman, 2013 ). This is especially true for higher education organisations that promote research and scholarly activities. Many European institutions have developed their own regulations for ethics by incorporating international codes (Getz, 1990 ). The lesser developed countries are trying to set up these committees to govern their academia and research. World Health Organization has stated that adhering to “ ethical principles … [is central and important]... in order to protect the dignity, rights and welfare of research participants ” (WHO, 2021 ). Ethical guidelines taught to students can help develop ethical researchers and members of society who uphold values of ethical principles in practice.
As the first European-wide consortium established to assist academic integrity (European Network for Academic Integrity – ENAI), we felt the importance of guiding those institutions and communities that are trying to teach, research, and include ethical principles by providing overarching understanding of ethical guidelines that may influence policy. Therefore, we set up an advisory working group within ENAI in 2018 to support matters related to ethics, ethical committees and assisting on ethics related teaching activities.
Upon preliminary review and discussion, the group found a disparity in understanding, practice and teaching approaches to ethical applications among peers. This became the premise for this research paper. We first carried out a literature survey to review and summarise existing ethical governance (with historical perspectives) and procedures that are already in place to guide researchers in different discipline areas. By doing so, we attempted to consolidate, document and provide important steps in a typical ethical application process with example procedures from different universities. Finally, we attempted to provide insights and findings from practical workshops carried out at the 5th International Conference Plagiarism across Europe and Beyond, in Mykolas Romeris University, Lithuania in 2019, focussing on:
• highlighting the basic needs of an ethical committee within an institution,
• discussing and sharing examples of a typical ethical approval process,
• providing guidelines on the ways to teach research ethics with some examples.
We believe this paper provides guidelines on preparing and training both researchers and research students in appropriately upholding ethical practices through ethical approval processes.
Background literature survey
Responsible research practice (RRP) is scrutinised by the aspects of ethical principles and professional standards (WHO’s Code of Conduct for responsible Research, 2017). The Singapore statement on research integrity (The Singapore Statement on Research integrity, 2010) has provided an internationally acceptable guidance for RRP. The statement is based on maintaining honesty, accountability, professional courtesy in all aspects of research and maintaining fairness during collaborations. In other words, it does not simply focus on the procedural part of the research, instead covers wider aspects of “integrity” beyond the operational aspects (Israel and Drenth, 2016 ).
Institutions should focus on providing ethical guidance based on principles and values reflecting upon all aspects/stages of research (from the funding application/project development stage upto or beyond project closing stage). Figure 1 summarizes the different aspects/stages of a typical research and highlights the needs of RRP in compliance with ethical governance at each stage with examples (the figure is based on Resnik, 2020 ; Žukauskas et al., 2018 ; Anderson, 2011 ; Fouka and Mantzorou, 2011 ).
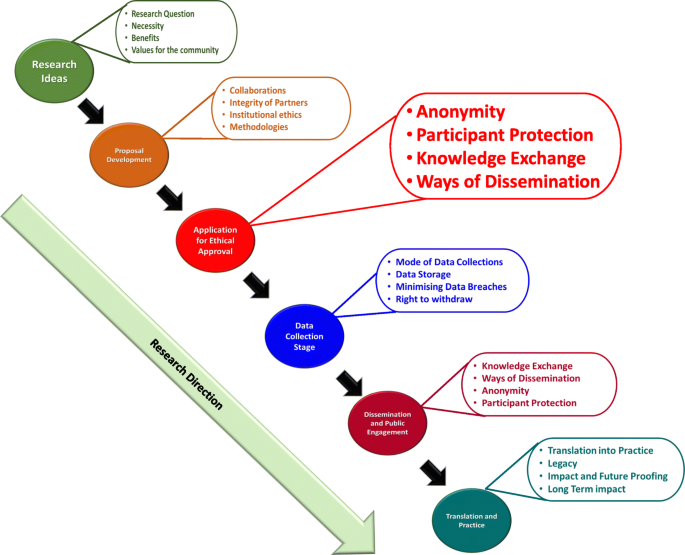
Summary of the enabling ethical governance at different stages of research. Note that it is imperative for researchers to proactively consider the ethical implications before, during and after the actual research process. The summary shows that RRP should be in line with ethical considerations even long before the ethical approval stage
Individual responsibilities to enhance RRP
As explained in Fig. 1 , a successfully governed research should consider ethics at the planning stages prior to research. Many international guidance are compatible in enforcing/recommending 14 different “responsibilities” that were first highlighted in the Singapore Statement (2010) for researchers to follow and achieve competency in RRP. In order to understand the purpose and the expectation of these ethical guidelines, we have carried out an initial literature survey on expected individual responsibilities. These are summarised in Table 1 .
By following these directives, researchers can carry out accountable research by maximising ethical self-governance whilst minimising misconducts. In our own experiences of working with many researchers, their focus usually revolves around ethical “clearance” rather than behaviour. In other words, they perceive this as a paper exercise rather than trying to “own” ethical behaviour in everything they do. Although the ethical principles and responsibilities are explicitly highlighted in the majority of international guidelines [such as UK’s Research Governance Policy (NICE, 2018 ), Australian Government’s National Statement on Ethical Conduct in Human Research (Difn website a - National Statement on Ethical Conduct in Human Research (NSECHR), 2018 ), the Singapore Statement (2010) etc.]; and the importance of holistic approach has been argued in ethical decision making, many researchers and/or institutions only focus on ethics linked to the procedural aspects.
Studies in the past have also highlighted inconsistencies in institutional guidelines pointing to the fact that these inconsistencies may hinder the predicted research progress (Desmond & Dierickx 2021 ; Alba et al., 2020 ; Dellaportas et al., 2014 ; Speight 2016 ). It may also be possible that these were and still are linked to the institutional perceptions/expectations or the pre-empting contextual conditions that are imposed by individual countries. In fact, it is interesting to note many research organisations and HE institutions establish their own policies based on these directives.
Research governance - origins, expectations and practices
Ethical governance in clinical medicine helps us by providing a structure for analysis and decision-making. By providing workable definitions of benefits and risks as well as the guidance for evaluating/balancing benefits over risks, it supports the researchers to protect the participants and the general population.
According to the definition given by National Institute of Clinical care Excellence, UK (NICE 2018 ), “ research governance can be defined as the broad range of regulations, principles and standards of good practice that ensure high quality research ”. As stated above, our literature-based research survey showed that most of the ethical definitions are basically evolved from the medical field and other disciplines have utilised these principles to develop their own ethical guidance. Interestingly, historical data show that the medical research has been “self-governed” or in other words implicated by the moral behaviour of individual researchers (Fox 2017 ; Shaw et al., 2005 ; Getz, 1990 ). For example, early human vaccination trials conducted in 1700s used the immediate family members as test subjects (Fox, 2017 ). Here the moral justification might have been the fact that the subjects who would have been at risk were either the scientists themselves or their immediate families but those who would reap the benefits from the vaccination were the general public/wider communities. However, according to the current ethical principles, this assumption is entirely not acceptable.
Historically, ambiguous decision-making and resultant incidences of research misconduct have led to the need for ethical research governance in as early as the 1940’s. For instance, the importance of an international governance was realised only after the World War II, when people were astonished to note the unethical research practices carried out by Nazi scientists. As a result of this, in 1947 the Nuremberg code was published. The code mainly focussed on the following:
Informed consent and further insisted the research involving humans should be based on prior animal work,
The anticipated benefits should outweigh the risk,
Research should be carried out only by qualified scientists must conduct research,
Avoiding physical and mental suffering and.
Avoiding human research that would result in which death or disability.
(Weindling, 2001 ).
Unfortunately, it was reported that many researchers in the USA and elsewhere considered the Nuremberg code as a document condemning the Nazi atrocities, rather than a code for ethical governance and therefore ignored these directives (Ghooi, 2011 ). It was only in 1964 that the World Medical Association published the Helsinki Declaration, which set the stage for ethical governance and the implementation of the Institutional Review Board (IRB) process (Shamoo and Irving, 1993 ). This declaration was based on Nuremberg code. In addition, the declaration also paved the way for enforcing research being conducted in accordance with these guidelines.
Incidentally, the focus on research/ethical governance gained its momentum in 1974. As a result of this, a report on ethical principles and guidelines for the protection of human subjects of research was published in 1979 (The Belmont Report, 1979 ). This report paved the way to the current forms of ethical governance in biomedical and behavioural research by providing guidance.
Since 1994, the WHO itself has been providing several guidance to health care policy-makers, researchers and other stakeholders detailing the key concepts in medical ethics. These are specific to applying ethical principles in global public health.
Likewise, World Organization for Animal Health (WOAH), and International Convention for the Protection of Animals (ICPA) provide guidance on animal welfare in research. Due to this continuous guidance, together with accepted practices, there are internationally established ethical guidelines to carry out medical research. Our literature survey further identified freely available guidance from independent organisations such as COPE (Committee of Publication Ethics) and ALLEA (All European Academics) which provide support for maintaining research ethics in other fields such as education, sociology, psychology etc. In reality, ethical governance is practiced differently in different countries. In the UK, there is a clinical excellence research governance, which oversees all NHS related medical research (Mulholland and Bell, 2005 ). Although, the governance in other disciplines is not entirely centralised, many research funding councils and organisations [such as UKRI (UK-Research and Innovation; BBSC (Biotechnology and Biological Sciences Research Council; MRC (Medical Research Council); EPSRC (Economic and Social Research Council)] provide ethical governance and expect institutional adherence and monitoring. They expect local institutional (i.e. university/institutional) research governance for day-to-day monitoring of the research conducted within the organisation and report back to these funding bodies, monthly or annually (Department of Health, 2005). Likewise, there are nationally coordinated/regulated ethics governing bodies such as the US Office for Human Research Protections (US-OHRP), National Institute of Health (NIH) and the Canadian Institutes for Health Research (CIHR) in the USA and Canada respectively (Mulholland and Bell, 2005 ). The OHRP in the USA formally reviews all research activities involving human subjects. On the other hand, in Canada, CIHR works with the Natural Sciences and Engineering Research Council (NSERC), and the Social Sciences and Humanities Research Council (SSHRC). They together have produced a Tri-Council Policy Statement (TCPS) (Stephenson et al., 2020 ) as ethical governance. All Canadian institutions are expected to adhere to this policy for conducting research. As for Australia, the research is governed by the Australian code for the responsible conduct of research (2008). It identifies the responsibilities of institutions and researchers in all areas of research. The code has been jointly developed by the National Health and Medical Research Council (NHMRC), the Australian Research Council (ARC) and Universities Australia (UA). This information is summarized in Table 2 .
Basic structure of an institutional ethical advisory committee (EAC)
The WHO published an article defining the basic concepts of an ethical advisory committee in 2009 (WHO, 2009 - see above). According to this, many countries have established research governance and monitor the ethical practice in research via national and/or regional review committees. The main aims of research ethics committees include reviewing the study proposals, trying to understand the justifications for human/animal use, weighing the merits and demerits of the usage (linking to risks vs. potential benefits) and ensuring the local, ethical guidelines are followed Difn website b - Enago academy Importance of Ethics Committees in Scholarly Research, 2020 ; Guide for Research Ethics - Council of Europe, 2014 ). Once the research has started, the committee needs to carry out periodic surveillance to ensure the institutional ethical norms are followed during and beyond the study. They may also be involved in setting up and/or reviewing the institutional policies.
For these aspects, IRB (or institutional ethical advisory committee - IEAC) is essential for local governance to enhance best practices. The advantage of an IRB/EEAC is that they understand the institutional conditions and can closely monitor the ongoing research, including any changes in research directions. On the other hand, the IRB may be overly supportive to accept applications, influenced by the local agenda for achieving research excellence, disregarding ethical issues (Kotecha et al., 2011 ; Kayser-Jones, 2003 ) or, they may be influenced by the financial interests in attracting external funding. In this respect, regional and national ethics committees are advantageous to ensure ethical practice. Due to their impartiality, they would provide greater consistency and legitimacy to the research (WHO, 2009 ). However, the ethical approval process of regional and national ethics committees would be time consuming, as they do not have the local knowledge.
As for membership in the IRBs, most of the guidelines [WHO, NICE, Council of Europe, (2012), European Commission - Facilitating Research Excellence in FP7 ( 2013 ) and OHRP] insist on having a variety of representations including experts in different fields of research, and non-experts with the understanding of local, national/international conflicts of interest. The former would be able to understand/clarify the procedural elements of the research in different fields; whilst the latter would help to make neutral and impartial decisions. These non-experts are usually not affiliated to the institution and consist of individuals representing the broader community (particularly those related to social, legal or cultural considerations). IRBs consisting of these varieties of representation would not only be in a position to understand the study procedures and their potential direct or indirect consequences for participants, but also be able to identify any community, cultural or religious implications of the study.
Understanding the subtle differences between ethics and morals
Interestingly, many ethical guidelines are based on society’s moral “beliefs” in such a way that the words “ethics”‘and “morals” are reciprocally used to define each other. However, there are several subtle differences between them and we have attempted to compare and contrast them herein. In the past, many authors have interchangeably used the words “morals”‘and “ethics”‘(Warwick, 2003 ; Kant, 2018 ; Hazard, GC (Jr)., 1994 , Larry, 1982 ). However, ethics is linked to rules governed by an external source such as codes of conduct in workplaces (Kuyare et al., 2014 ). In contrast, morals refer to an individual’s own principles regarding right and wrong. Quinn ( 2011 ) defines morality as “ rules of conduct describing what people ought and ought not to do in various situations … ” while ethics is “... the philosophical study of morality, a rational examination into people’s moral beliefs and behaviours ”. For instance, in a case of parents demanding that schools overturn a ban on use of corporal punishment of children by schools and teachers (Children’s Rights Alliance for England, 2005 ), the parents believed that teachers should assume the role of parent in schools and use corporal or physical punishment for children who misbehaved. This stemmed from their beliefs and what they felt were motivated by “beliefs of individuals or groups”. For example, recent media highlights about some parents opposing LGBT (Lesbian, Gay, Bisexual, and Transgender) education to their children (BBC News, 2019 ). One parent argued, “Teaching young children about LGBT at a very early stage is ‘morally’ wrong”. She argued “let them learn by themselves as they grow”. This behaviour is linked to and governed by the morals of an ethnic community. Thus, morals are linked to the “beliefs of individuals or group”. However, when it comes to the LGBT rights these are based on ethical principles of that society and governed by law of the land. However, the rights of children to be protected from “inhuman and degrading” treatment is based on the ethical principles of the society and governed by law of the land. Individuals, especially those who are working in medical or judicial professions have to follow an ethical code laid down by their profession, regardless of their own feelings, time or preferences. For instance, a lawyer is expected to follow the professional ethics and represent a defendant, despite the fact that his morals indicate the defendant is guilty.
In fact, we as a group could not find many scholarly articles clearly comparing or contrasting ethics with morals. However, a table presented by Surbhi ( 2015 ) (Difn website c ) tries to differentiate these two terms (see Table 3 ).
Although Table 3 gives some insight on the differences between these two terms, in practice many use these terms as loosely as possible mainly because of their ambiguity. As a group focussed on the application of these principles, we would recommend to use the term “ethics” and avoid “morals” in research and academia.
Based on the literature survey carried out, we were able to identify the following gaps:
there is some disparity in existing literature on the importance of ethical guidelines in research
there is a lack of consensus on what code of conduct should be followed, where it should be derived from and how it should be implemented
The mission of ENAI’s ethical advisory working group
The Ethical Advisory Working Group of ENAI was established in 2018 to promote ethical code of conduct/practice amongst higher educational organisations within Europe and beyond (European Network for Academic Integrity, 2018 ). We aim to provide unbiased advice and consultancy on embedding ethical principles within all types of academic, research and public engagement activities. Our main objective is to promote ethical principles and share good practice in this field. This advisory group aims to standardise ethical norms and to offer strategic support to activities including (but not exclusive to):
● rendering advice and assistance to develop institutional ethical committees and their regulations in member institutions,
● sharing good practice in research and academic ethics,
● acting as a critical guide to institutional review processes, assisting them to maintain/achieve ethical standards,
● collaborating with similar bodies in establishing collegiate partnerships to enhance awareness and practice in this field,
● providing support within and outside ENAI to develop materials to enhance teaching activities in this field,
● organising training for students and early-career researchers about ethical behaviours in form of lectures, seminars, debates and webinars,
● enhancing research and dissemination of the findings in matters and topics related to ethics.
The following sections focus on our suggestions based on collective experiences, review of literature provided in earlier sections and workshop feedback collected:
a) basic needs of an ethical committee within an institution;
b) a typical ethical approval process (with examples from three different universities); and
c) the ways to obtain informed consent with some examples. This would give advice on preparing and training both researchers and research students in appropriately upholding ethical practices through ethical approval processes.
Setting up an institutional ethical committee (ECs)
Institutional Ethical Committees (ECs) are essential to govern every aspect of the activities undertaken by that institute. With regards to higher educational organisations, this is vital to establish ethical behaviour for students and staff to impart research, education and scholarly activities (or everything) they do. These committees should be knowledgeable about international laws relating to different fields of studies (such as science, medicine, business, finance, law, and social sciences). The advantages and disadvantages of institutional, subject specific or common (statutory) ECs are summarised in Fig. 2 . Some institutions have developed individual ECs linked to specific fields (or subject areas) whilst others have one institutional committee that overlooks the entire ethical behaviour and approval process. There is no clear preference between the two as both have their own advantages and disadvantages (see Fig. 2 ). Subject specific ECs are attractive to medical, law and business provisions, as it is perceived the members within respective committees would be able to understand the subject and therefore comprehend the need of the proposed research/activity (Kadam, 2012 ; Schnyder et al., 2018 ). However, others argue, due to this “ specificity ”, the committee would fail to forecast the wider implications of that application. On the other hand, university-wide ECs would look into the wider implications. Yet they find it difficult to understand the purpose and the specific applications of that research. Not everyone understands dynamics of all types of research methodologies, data collection, etc., and therefore there might be a chance of a proposal being rejected merely because the EC could not understand the research applications (Getz, 1990 ).

Summary of advantages and disadvantages of three different forms of ethical committees
[N/B for Fig. 2 : Examples of different types of ethical application procedures and forms used were discussed with the workshop attendees to enhance their understanding of the differences. GDPR = General Data Protection Regulation].
Although we recommend a designated EC with relevant professional, academic and ethical expertise to deal with particular types of applications, the membership (of any EC) should include some non-experts who would represent the wider community (see above). Having some non-experts in EC would not only help the researchers to consider explaining their research in layperson’s terms (by thinking outside the box) but also would ensure efficiency without compromising participants/animal safety. They may even help to address the common ethical issues outside research culture. Some UK universities usually offer this membership to a clergy, councillor or a parliamentarian who does not have any links to the institutions. Most importantly, it is vital for any EC members to undertake further training in addition to previous experience in the relevant field of research ethics.
Another issue that raises concerns is multi-centre research, involving several institutions, where institutionalised ethical approvals are needed from each partner. In some cases, such as clinical research within the UK, a common statutory EC called National Health Services (NHS) Research Ethics Committee (NREC) is in place to cover research ethics involving all partner institutions (NHS, 2018 ). The process of obtaining approval from this type of EC takes time, therefore advanced planning is needed.
Ethics approval forms and process
During the workshop, we discussed some anonymised application forms obtained from open-access sources for qualitative and quantitative research as examples. Considering research ethics, for the purpose of understanding, we arbitrarily divided this in two categories; research based on (a) quantitative and (b) qualitative methodologies. As their name suggests their research approach is extremely different from each other. The discussion elicited how ECs devise different types of ethical application form/questions. As for qualitative research, these are often conducted as “face-to-face” interviews, which would have implications on volunteer anonymity.
Furthermore, discussions posited when the interviews are replaced by on-line surveys, they have to be administered through registered university staff to maintain confidentiality. This becomes difficult when the research is a multi-centre study. These types of issues are also common in medical research regarding participants’ anonymity, confidentially, and above all their right to withdraw consent to be involved in research.
Storing and protecting data collected in the process of the study is also a point of consideration when applying for approval.
Finally, the ethical processes of invasive (involving human/animals) and non-invasive research (questionnaire based) may slightly differ from one another. Following research areas are considered as investigations that need ethical approval:
research that involves human participants (see below)
use of the ‘products’ of human participants (see below)
work that potentially impacts on humans (see below)
research that involves animals
In addition, it is important to provide a disclaimer even if an ethical approval is deemed unnecessary. Following word cloud (Fig. 3 ) shows the important variables that need to be considered at the brainstorming stage before an ethical application. It is worth noting the importance of proactive planning predicting the “unexpected” during different phases of a research project (such as planning, execution, publication, and future directions). Some applications (such as working with vulnerable individuals or children) will require safety protection clearance (such as DBS - Disclosure and Barring Service, commonly obtained from the local police). Please see section on Research involving Humans - Informed consents for further discussions.

Examples of important variables that need to be considered for an ethical approval
It is also imperative to report or re-apply for ethical approval for any minor or major post-approval changes to original proposals made. In case of methodological changes, evidence of risk assessments for changes and/or COSHH (Control of Substances Hazardous to Health Regulations) should also be given. Likewise, any new collaborative partners or removal of researchers should also be notified to the IEAC.
Other findings include:
in case of complete changes in the project, the research must be stopped and new approval should be seeked,
in case of noticing any adverse effects to project participants (human or non-human), these should also be notified to the committee for appropriate clearance to continue the work, and
the completion of the project must also be notified with the indication whether the researchers may restart the project at a later stage.
Research involving humans - informed consents
While discussing research involving humans and based on literature review, findings highlight the human subjects/volunteers must willingly participate in research after being adequately informed about the project. Therefore, research involving humans and animals takes precedence in obtaining ethical clearance and its strict adherence, one of which is providing a participant information sheet/leaflet. This sheet should contain a full explanation about the research that is being carried out and be given out in lay-person’s terms in writing (Manti and Licari 2018 ; Hardicre 2014 ). Measures should also be in place to explain and clarify any doubts from the participants. In addition, there should be a clear statement on how the participants’ anonymity is protected. We provide below some example questions below to help the researchers to write this participant information sheet:
What is the purpose of the study?
Why have they been chosen?
What will happen if they take part?
What do they have to do?
What happens when the research stops?
What if something goes wrong?
What will happen to the results of the research study?
Will taking part be kept confidential?
How to handle “vulnerable” participants?
How to mitigate risks to participants?
Many institutional ethics committees expect the researchers to produce a FAQ (frequently asked questions) in addition to the information about research. Most importantly, the researchers also need to provide an informed consent form, which should be signed by each human participant. The five elements identified that are needed to be considered for an informed consent statement are summarized in Fig. 4 below (slightly modified from the Federal Policy for the Protection of Human Subjects ( 2018 ) - Diffn website c ).
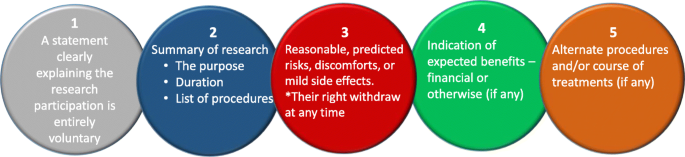
Five basic elements to consider for an informed consent [figure adapted from Diffn website c ]
The informed consent form should always contain a clause for the participant to withdraw their consent at any time. Should this happen all the data from that participant should be eliminated from the study without affecting their anonymity.
Typical research ethics approval process
In this section, we provide an example flow chart explaining how researchers may choose the appropriate application and process, as highlighted in Fig. 5 . However, it is imperative to note here that these are examples only and some institutions may have one unified application with separate sections to demarcate qualitative and quantitative research criteria.
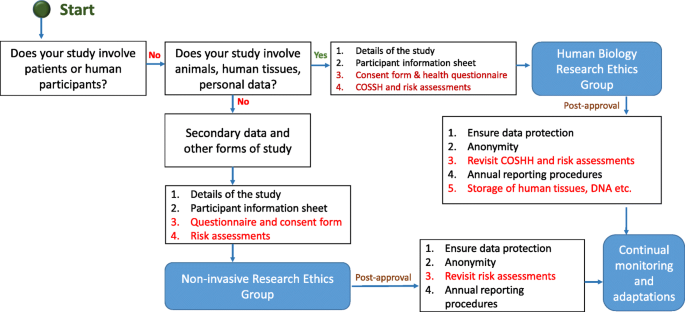
Typical ethical approval processes for quantitative and qualitative research. [N/B for Fig. 5 - This simplified flow chart shows that fundamental process for invasive and non-invasive EC application is same, the routes and the requirements for additional information are slightly different]
Once the ethical application is submitted, the EC should ensure a clear approval procedure with distinctly defined timeline. An example flow chart showing the procedure for an ethical approval was obtained from University of Leicester as open-access. This is presented in Fig. 6 . Further examples of the ethical approval process and governance were discussed in the workshop.
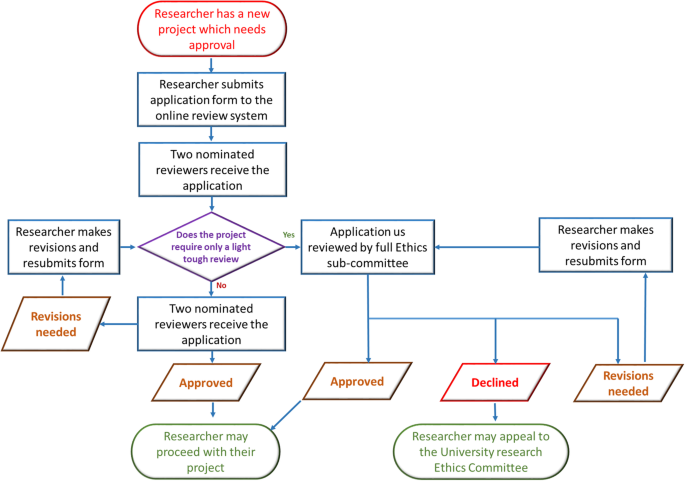
An example ethical approval procedures conducted within University of Leicester (Figure obtained from the University of Leicester research pages - Difn website d - open access)
Strategies for ethics educations for students
Student education on the importance of ethics and ethical behaviour in research and scholarly activities is extremely essential. Literature posits in the area of medical research that many universities are incorporating ethics in post-graduate degrees but when it comes to undergraduate degrees, there is less appetite to deliver modules or even lectures focussing on research ethics (Seymour et al., 2004 ; Willison and O’Regan, 2007 ). This may be due to the fact that undergraduate degree structure does not really focus on research (DePasse et al., 2016 ). However, as Orr ( 2018 ) suggested, institutions should focus more on educating all students about ethics/ethical behaviour and their importance in research, than enforcing punitive measures for unethical behaviour. Therefore, as an advisory committee, and based on our preliminary literature survey and workshop results, we strongly recommend incorporating ethical education within undergraduate curriculum. Looking at those institutions which focus on ethical education for both under-and postgraduate courses, their approaches are either (a) a lecture-based delivery, (b) case study based approach or (c) a combined delivery starting with a lecture on basic principles of ethics followed by generating a debate based discussion using interesting case studies. The combined method seems much more effective than the other two as per our findings as explained next.
As many academics who have been involved in teaching ethics and/or research ethics agree, the underlying principles of ethics is often perceived as a boring subject. Therefore, lecture-based delivery may not be suitable. On the other hand, a debate based approach, though attractive and instantly generates student interest, cannot be effective without students understanding the underlying basic principles. In addition, when selecting case studies, it would be advisable to choose cases addressing all different types of ethical dilemmas. As an advisory group within ENAI, we are in the process of collating supporting materials to help to develop institutional policies, creating advisory documents to help in obtaining ethical approvals, and teaching materials to enhance debate-based lesson plans that can be used by the member and other institutions.
Concluding remarks
In summary, our literature survey and workshop findings highlight that researchers should accept that ethics underpins everything we do, especially in research. Although ethical approval is tedious, it is an imperative process in which proactive thinking is essential to identify ethical issues that might affect the project. Our findings further lead us to state that the ethical approval process differs from institution to institution and we strongly recommend the researchers to follow the institutional guidelines and their underlying ethical principles. The ENAI workshop in Vilnius highlighted the importance of ethical governance by establishing ECs, discussed different types of ECs and procedures with some examples and highlighted the importance of student education to impart ethical culture within research communities, an area that needs further study as future scope.
Declarations
The manuscript was entirely written by the corresponding author with contributions from co-authors who have also taken part in the delivery of the workshop. Authors confirm that the data supporting the findings of this study are available within the article. We can also confirm that there are no potential competing interests with other organisations.
Availability of data and materials
Authors confirm that the data supporting the findings of this study are available within the article.
Abbreviations
ALL European academics
Australian research council
Biotechnology and biological sciences research council
Canadian institutes for health research
Committee of publication ethics
Ethical committee
European network of academic integrity
Economic and social research council
International convention for the protection of animals
institutional ethical advisory committee
Institutional review board
Immaculata university of Pennsylvania
Lesbian, gay, bisexual, and transgender
Medical research council)
National health services
National health services nih national institute of health (NIH)
National institute of clinical care excellence
National health and medical research council
Natural sciences and engineering research council
National research ethics committee
National statement on ethical conduct in human research
Responsible research practice
Social sciences and humanities research council
Tri-council policy statement
World Organization for animal health
Universities Australia
UK-research and innovation
US office for human research protections
Alba S, Lenglet A, Verdonck K, Roth J, Patil R, Mendoza W, Juvekar S, Rumisha SF (2020) Bridging research integrity and global health epidemiology (BRIDGE) guidelines: explanation and elaboration. BMJ Glob Health 5(10):e003237. https://doi.org/10.1136/bmjgh-2020-003237
Article Google Scholar
Anderson MS (2011) Research misconduct and misbehaviour. In: Bertram Gallant T (ed) Creating the ethical academy: a systems approach to understanding misconduct and empowering change in higher education. Routledge, pp 83–96
BBC News. (2019). Birmingham school LGBT LESSONS PROTEST investigated. March 8, 2019. Retrieved February 14, 2021, available online. URL: https://www.bbc.com/news/uk-england-birmingham-47498446
Children’s Rights Alliance for England. (2005). R (Williamson and others) v Secretary of State for Education and Employment. Session 2004–05. [2005] UKHL 15. Available Online. URL: http://www.crae.org.uk/media/33624/R-Williamson-and-others-v-Secretary-of-State-for-Education-and-Employment.pdf
Council of Europe. (2014). Texts of the Council of Europe on bioethical matters. Available Online. https://www.coe.int/t/dg3/healthbioethic/Texts_and_documents/INF_2014_5_vol_II_textes_%20CoE_%20bio%C3%A9thique_E%20(2).pdf
Dellaportas S, Kanapathippillai S, Khan, A and Leung, P. (2014). Ethics education in the Australian accounting curriculum: a longitudinal study examining barriers and enablers. 362–382. Available Online. URL: https://doi.org/10.1080/09639284.2014.930694 , 23, 4, 362, 382
DePasse JM, Palumbo MA, Eberson CP, Daniels AH (2016) Academic characteristics of orthopaedic surgery residency applicants from 2007 to 2014. JBJS 98(9):788–795. https://doi.org/10.2106/JBJS.15.00222
Desmond H, Dierickx K (2021) Research integrity codes of conduct in Europe: understanding the divergences. https://doi.org/10.1111/bioe.12851
Difn website a - National Statement on Ethical Conduct in Human Research (NSECHR). (2018). Available Online. URL: https://www.nhmrc.gov.au/about-us/publications/australian-code-responsible-conduct-research-2018
Difn website b - Enago academy Importance of Ethics Committees in Scholarly Research (2020, October 26). Available online. URL: https://www.enago.com/academy/importance-of-ethics-committees-in-scholarly-research/
Difn website c - Ethics vs Morals - Difference and Comparison. Retrieved July 14, 2020. Available online. URL: https://www.diffen.com/difference/Ethics_vs_Morals
Difn website d - University of Leicester. (2015). Staff ethics approval flowchart. May 1, 2015. Retrieved July 14, 2020. Available Online. URL: https://www2.le.ac.uk/institution/ethics/images/ethics-approval-flowchart/view
European Commission - Facilitating Research Excellence in FP7 (2013) https://ec.europa.eu/research/participants/data/ref/fp7/89888/ethics-for-researchers_en.pdf
European Network for Academic Integrity. (2018). Ethical advisory group. Retrieved February 14, 2021. Available online. URL: http://www.academicintegrity.eu/wp/wg-ethical/
Federal Policy for the Protection of Human Subjects. (2018). Retrieved February 14, 2021. Available Online. URL: https://www.federalregister.gov/documents/2017/01/19/2017-01058/federal-policy-for-the-protection-of-human-subjects#p-855
Flite, CA and Harman, LB. (2013). Code of ethics: principles for ethical leadership Perspect Health Inf Mana; 10(winter): 1d. PMID: 23346028
Fouka G, Mantzorou M (2011) What are the major ethical issues in conducting research? Is there a conflict between the research ethics and the nature of nursing. Health Sci J 5(1) Available Online. URL: https://www.hsj.gr/medicine/what-are-the-major-ethical-issues-in-conducting-research-is-there-a-conflict-between-the-research-ethics-and-the-nature-of-nursing.php?aid=3485
Fox G (2017) History and ethical principles. The University of Miami and the Collaborative Institutional Training Initiative (CITI) Program URL https://silo.tips/download/chapter-1-history-and-ethical-principles # (Available Online)
Getz KA (1990) International codes of conduct: An analysis of ethical reasoning. J Bus Ethics 9(7):567–577
Ghooi RB (2011) The nuremberg code–a critique. Perspect Clin Res 2(2):72–76. https://doi.org/10.4103/2229-3485.80371
Hardicre, J. (2014) Valid informed consent in research: an introduction Br J Nurs 23(11). https://doi.org/10.12968/bjon.2014.23.11.564 , 567
Hazard, GC (Jr). (1994). Law, morals, and ethics. Yale law school legal scholarship repository. Faculty Scholarship Series. Yale University. Available Online. URL: https://digitalcommons.law.yale.edu/cgi/viewcontent.cgi?referer=https://www.google.com/&httpsredir=1&article=3322&context=fss_papers
Israel, M., & Drenth, P. (2016). Research integrity: perspectives from Australia and Netherlands. In T. Bretag (Ed.), Handbook of academic integrity (pp. 789–808). Springer, Singapore. https://doi.org/10.1007/978-981-287-098-8_64
Kadam R (2012) Proactive role for ethics committees. Indian J Med Ethics 9(3):216. https://doi.org/10.20529/IJME.2012.072
Kant I (2018) The metaphysics of morals. Cambridge University Press, UK https://doi.org/10.1017/9781316091388
Kayser-Jones J (2003) Continuing to conduct research in nursing homes despite controversial findings: reflections by a research scientist. Qual Health Res 13(1):114–128. https://doi.org/10.1177/1049732302239414
Kotecha JA, Manca D, Lambert-Lanning A, Keshavjee K, Drummond N, Godwin M, Greiver M, Putnam W, Lussier M-T, Birtwhistle R (2011) Ethics and privacy issues of a practice-based surveillance system: need for a national-level institutional research ethics board and consent standards. Can Fam physician 57(10):1165–1173. https://europepmc.org/article/pmc/pmc3192088
Kuyare, MS., Taur, SR., Thatte, U. (2014). Establishing institutional ethics committees: challenges and solutions–a review of the literature. Indian J Med Ethics. https://doi.org/10.20529/IJME.2014.047
LaFollette, H. (2007). Ethics in practice (3rd edition). Blackwell
Larry RC (1982) The teaching of ethics and moral values in teaching. J High Educ 53(3):296–306. https://doi.org/10.1080/00221546.1982.11780455
Manti S, Licari A (2018) How to obtain informed consent for research. Breathe (Sheff) 14(2):145–152. https://doi.org/10.1183/20734735.001918
Mulholland MW, Bell J (2005) Research Governance and Research Funding in the USA: What the academic surgeon needs to know. J R Soc Med 98(11):496–502. https://doi.org/10.1258/jrsm.98.11.496
National Institute of Health (NIH) Ethics in Clinical Research. n.d. Available Online. URL: https://clinicalcenter.nih.gov/recruit/ethics.html
NHS (2018) Flagged Research Ethics Committees. Retrieved February 14, 2021. Available online. URL: https://www.hra.nhs.uk/about-us/committees-and-services/res-and-recs/flagged-research-ethics-committees/
NICE (2018) Research governance policy. Retrieved February 14, 2021. Available online. URL: https://www.nice.org.uk/Media/Default/About/what-we-do/science-policy-and-research/research-governance-policy.pdf
Orr, J. (2018). Developing a campus academic integrity education seminar. J Acad Ethics 16(3), 195–209. https://doi.org/10.1007/s10805-018-9304-7
Quinn, M. (2011). Introduction to Ethics. Ethics for an Information Age. 4th Ed. Ch 2. 53–108. Pearson. UK
Resnik. (2020). What is ethics in Research & why is it Important? Available Online. URL: https://www.niehs.nih.gov/research/resources/bioethics/whatis/index.cfm
Schnyder S, Starring H, Fury M, Mora A, Leonardi C, Dasa V (2018) The formation of a medical student research committee and its impact on involvement in departmental research. Med Educ Online 23(1):1. https://doi.org/10.1080/10872981.2018.1424449
Seymour E, Hunter AB, Laursen SL, DeAntoni T (2004) Establishing the benefits of research experiences for undergraduates in the sciences: first findings from a three-year study. Sci Educ 88(4):493–534. https://doi.org/10.1002/sce.10131
Shamoo AE, Irving DN (1993) Accountability in research using persons with mental illness. Account Res 3(1):1–17. https://doi.org/10.1080/08989629308573826
Shaw, S., Boynton, PM., and Greenhalgh, T. (2005). Research governance: where did it come from, what does it mean? Research governance framework for health and social care, 2nd ed. London: Department of Health. https://doi.org/10.1258/jrsm.98.11.496 , 98, 11, 496, 502
Book Google Scholar
Speight, JG. (2016) Ethics in the university |DOI: https://doi.org/10.1002/9781119346449 scrivener publishing LLC
Stephenson GK, Jones GA, Fick E, Begin-Caouette O, Taiyeb A, Metcalfe A (2020) What’s the protocol? Canadian university research ethics boards and variations in implementing tri-Council policy. Can J Higher Educ 50(1)1): 68–81
Surbhi, S. (2015). Difference between morals and ethics [weblog]. March 25, 2015. Retrieved February 14, 2021. Available Online. URL: http://keydifferences.com/difference-between-morals-and-ethics.html
The Belmont Report (1979). Ethical Principles and Guidelines for the Protection of Human Subjects of Research. The National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. Retrieved February 14, 2021. Available online. URL: https://www.hhs.gov/ohrp/sites/default/files/the-belmont-report-508c_FINAL.pdf
The Singapore Statement on Research Integrity. (2020). Nicholas Steneck and Tony Mayer, Co-chairs, 2nd World Conference on Research Integrity; Melissa Anderson, Chair, Organizing Committee, 3rd World Conference on Research Integrity. Retrieved February 14, 2021. Available online. URL: https://wcrif.org/documents/327-singapore-statement-a4size/file
Warwick K (2003) Cyborg morals, cyborg values, cyborg ethics. Ethics Inf Technol 5(3):131–137. https://doi.org/10.1023/B:ETIN.0000006870.65865.cf
Weindling P (2001) The origins of informed consent: the international scientific commission on medical war crimes, and the Nuremberg code. Bull Hist Med 75(1):37–71. https://doi.org/10.1353/bhm.2001.0049
WHO. (2009). Research ethics committees Basic concepts for capacity-building. Retrieved February 14, 2021. Available online. URL: https://www.who.int/ethics/Ethics_basic_concepts_ENG.pdf
WHO. (2021). Chronological list of publications. Retrieved February 14, 2021. Available online. URL: https://www.who.int/ethics/publications/year/en/
Willison, J. and O’Regan, K. (2007). Commonly known, commonly not known, totally unknown: a framework for students becoming researchers. High Educ Res Dev 26(4). 393–409. https://doi.org/10.1080/07294360701658609
Žukauskas P, Vveinhardt J, and Andriukaitienė R. (2018). Research Ethics In book: Management Culture and Corporate Social Responsibility Eds Jolita Vveinhardt IntechOpenEditors DOI: https://doi.org/10.5772/intechopen.70629 , 2018
Download references
Acknowledgements
Authors wish to thank the organising committee of the 5th international conference named plagiarism across Europe and beyond, in Vilnius, Lithuania for accepting this paper to be presented in the conference.
Not applicable as this is an independent study, which is not funded by any internal or external bodies.
Author information
Authors and affiliations.
School of Human Sciences, University of Derby, DE22 1, Derby, GB, UK
Shivadas Sivasubramaniam
Department of Informatics, Mendel University in Brno, Zemědělská, 1665, Brno, Czechia
Dita Henek Dlabolová
Centre for Academic Integrity in the UAE, Faculty of Engineering & Information Sciences, University of Wollongong in Dubai, Dubai, UAE
Veronika Kralikova & Zeenath Reza Khan
You can also search for this author in PubMed Google Scholar
Contributions
The manuscript was entirely written by the corresponding author with contributions from co-authors who have equally contributed to presentation of this paper in the 5th international conference named plagiarism across Europe and beyond, in Vilnius, Lithuania. Authors have equally contributed for the information collection, which were then summarised as narrative explanations by the Corresponding author and Dr. Zeenath Reza Khan. Then checked and verified by Dr. Dlabolova and Ms. Králíková. The author(s) read and approved the final manuscript.
Corresponding author
Correspondence to Shivadas Sivasubramaniam .
Ethics declarations
Competing interests.
We can also confirm that there are no potential competing interest with other organisations.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Sivasubramaniam, S., Dlabolová, D.H., Kralikova, V. et al. Assisting you to advance with ethics in research: an introduction to ethical governance and application procedures. Int J Educ Integr 17 , 14 (2021). https://doi.org/10.1007/s40979-021-00078-6
Download citation
Received : 17 July 2020
Accepted : 25 April 2021
Published : 13 July 2021
DOI : https://doi.org/10.1007/s40979-021-00078-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Higher education
- Ethical codes
- Ethics committee
- Post-secondary education
- Institutional policies
- Research ethics

International Journal for Educational Integrity
ISSN: 1833-2595
- General enquiries: [email protected]
What is Research Ethics?
Originally published 1999-2013 at Resources for Research Ethics Education, a web project directed by Michael Kalichman, Ph.D., and Dena Plemmons, Ph.D., from the University of California-San Diego Research Ethics Program and the San Diego Research Ethics Consortium. Republished with permission.
See Also: Teaching Research Ethics: Why Teach?
Research Ethics is defined here to be the ethics of the planning, conduct, and reporting of research.
It is clear that research ethics should include:
- Protections of human and animal subjects
However, not all researchers use human or animal subjects, nor are the ethical dimensions of research confined solely to protections for research subjects. Other ethical challenges are rooted in many dimensions of research, including the:
- Collection, use, and interpretation of research data
- Methods for reporting and reviewing research plans or findings
- Relationships among researchers with one another
- Relationships between researchers and those that will be affected by their research
- Means for responding to misunderstandings, disputes, or misconduct
- Options for promoting ethical conduct in research
For the purpose of this online resource, the domain of research ethics is intended to include nothing less than the fostering of research that protects the interests of the public, the subjects of research, and the researchers themselves.
Important Ethical Distinctions
In discussing or teaching research ethics, it is important to keep some basic distinctions in mind.
Prescriptive vs. descriptive claims
It is important not to confuse moral claims about how people ought to behave with descriptive claims about how they in fact do behave. From the fact that gift authorship or signing off on unreviewed data may be “common practice” in some contexts, it doesn’t follow that they are morally or professionally justified. Nor is morality to be confused with the moral beliefs or ethical codes that a given group or society holds (how some group thinks people should live). A belief in segregation is not morally justified simply because it is widely held by a group of people or given society. Philosophers term this distinction between prescriptive and descriptive claims the “is-ought distinction.”
Law vs. morality
A second important distinction is that between morality and the law. The law may or may not conform to the demands of ethics (Kagan, 1998). To take a contemporary example: many believe that the law prohibiting federally funded stem cell research is objectionable on moral (as well as scientific) grounds, i.e., that such research can save lives and prevent much human misery. History is full of examples of bad laws, that is laws now regarded as morally unjustifiable, e.g., the laws of apartheid, laws prohibiting women from voting or inter-racial couples from marrying.
It is also helpful to distinguish between two different levels of discussion (or two different kinds of ethical questions): first-order or “ground-level” questions and second-order questions.
First-order questions
First-order moral questions concern what we should do. Such questions may be very general or quite specific. One might ask whether the tradition of “senior” authorship should be defended and preserved or, more generally, what are the principles that should go into deciding the issue of senior authorship. Such questions and the substantive proposals regarding how to answer them belong to the domain of what moral philosophers call “normative ethics.”
Second-order questions
Second-order moral questions concern the nature and purpose of morality itself. When someone claims that falsifying data is wrong, what exactly is the standing of this claim? What exactly does the word “wrong” mean in the conduct of scientific research? And what are we doing when we make claims about right and wrong, scientific integrity and research misconduct? These second-order questions are quite different from the ground-level questions about how to conduct one’s private or professional life raised above. They concern the nature of morality rather than its content, i.e., what acts are required, permitted or prohibited. This is the domain of what moral philosophers call “metaethics” (Kagan, 1998).
Ways to Approach Ethics
Each of these approaches provides moral principles and ways of thinking about the responsibilities, duties and obligations of moral life. Individually and jointly, they can provide practical guidance in ethical decision-making.
Deontological ethics
One of the most influential and familiar approaches to ethics is deontological ethics, associated with Immanuel Kant (1742-1804). Deontological ethics hold certain acts as right or wrong in themselves, e.g., promise breaking or lying. So, for example, in the context of research, fraud, plagiarism and misrepresentation are regarded as morally wrong in themselves, not simply because they (tend to) have bad consequences. The deontological approach is generally grounded in a single fundamental principle: Act as you would wish others to act towards you OR always treat persons as an end, never as a means to an end.
From such central principles are derived rules or guidelines for what is permitted, required and prohibited. Objections to principle-based or deontological ethics include the difficulty of applying highly general principles to specific cases, e.g.: Does treating persons as ends rule out physician-assisted suicide, or require it? Deontological ethics is generally contrasted to consequentialist ethics (Honderich, 1995).
Consequentialist ethics
According to consequentialist approaches, the rightness or wrongness of an action depends solely on its consequences. One should act in such a way as to bring about the best state of affairs, where the best state of affairs may be understood in various ways, e.g., as the greatest happiness for the greatest number of people, maximizing pleasure and minimizing pain or maximizing the satisfaction of preferences. A theory such as Utilitarianism (with its roots in the work of Jeremy Bentham and John Stuart Mill) is generally taken as the paradigm example of consequentialism. Objections to consequentialist ethics tend to focus on its willingness to regard individual rights and values as “negotiable.” So, for example, most people would regard murder as wrong independently of the fact that killing one person might allow several others to be saved (the infamous sacrifice of an ailing patient to provide organs for several other needy patients). Similarly, widespread moral opinion holds certain values important (integrity, justice) not only because they generally lead to good outcomes, but in and of themselves.
Virtue ethics
Virtue ethics focuses on moral character rather than action and behavior considered in isolation. Central to this approach is the question what ought we (as individuals, as scientists, as physicians) to be rather than simply what we ought to do. The emphasis here is on inner states, that is, moral dispositions and habits such as courage or a developed sense of personal integrity. Virtue ethics can be a useful approach in the context of RCR and professional ethics, emphasizing the importance of moral virtues such as compassion, honesty, and respect. This approach has also a great deal to offer in discussions of bioethical issues where a traditional emphasis on rights and abstract principles frequently results in polarized, stalled discussions (e.g., abortion debates contrasting the rights of the mother against the rights of the fetus).
An ethics of care
The term “ethics of care” grows out of the work of Carol Gilligan, whose empirical work in moral psychology claimed to discover a “different voice,” a mode of moral thinking distinct from principle-based moral thinking (e.g., the theories of Kant and Mill). An ethics of care stresses compassion and empathetic understanding, virtues Gilligan associated with traditional care-giving roles, especially those of women.
This approach differs from traditional moral theories in two important ways. First, it assumes that it is the connections between persons, e.g., lab teams, colleagues, parents and children, student and mentor, not merely the rights and obligations of discrete individuals that matter. The moral world, on this view, is best seen not as the interaction of discrete individuals, each with his or her own interests and rights, but as an interrelated web of obligations and commitment. We interact, much of the time, not as private individuals, but as members of families, couples, institutions, research groups, a given profession and so on. Second, these human relationships, including relationships of dependency, play a crucial role on this account in determining what our moral obligations and responsibilities are. So, for example, individuals have special responsibilities to care for their children, students, patients, and research subjects.
An ethics of care is thus particularly useful in discussing human and animal subjects research, issues of informed consent, and the treatment of vulnerable populations such as children, the infirm or the ill.
Casuistry or case study approaches
The case study approach begins from real or hypothetical cases. Its objective is to identify the intuitively plausible principles that should be taken into account in resolving the issues at hand. The case study approach then proceeds to critically evaluate those principles. In discussing whistle-blowing, for example, a good starting point is with recent cases of research misconduct, seeking to identify and evaluate principles such as a commitment to the integrity of science, protecting privacy, or avoiding false or unsubstantiated charges. In the context of RCR instruction, case studies provide one of the most interesting and effective approaches to developing sensitivity to ethical issues and to honing ethical decision-making skills.
Strictly speaking, casuistry is more properly understood as a method for doing ethics rather than as itself an ethical theory. However, casuistry is not wholly unconnected to ethical theory. The need for a basis upon which to evaluate competing principles, e.g., the importance of the well-being of an individual patient vs. a concern for just allocation of scarce medical resources, makes ethical theory relevant even with case study approaches.
Applied ethics
Applied ethics is a branch of normative ethics. It deals with practical questions particularly in relation to the professions. Perhaps the best known area of applied ethics is bioethics, which deals with ethical questions arising in medicine and the biological sciences, e.g., questions concerning the application of new areas of technology (stem cells, cloning, genetic screening, nanotechnology, etc.), end of life issues, organ transplants, and just distribution of healthcare. Training in responsible conduct of research or “research ethics” is merely one among various forms of professional ethics that have come to prominence since the 1960s. Worth noting, however, is that concern with professional ethics is not new, as ancient codes such as the Hippocratic Oath and guild standards attest (Singer, 1986).
Research Ethics
- Adams, D., Pimple, K.D. (2005). Research Misconduct and Crime: Lessons from Criminal Science on Preventing Misconduct and Promoting Integrity. Accountability in Research, 12 (3): 225-240.
- Anderson, M.S., Horn, A.S., Risbey, K.R., Ronning, E.A., De Vries, R., Martinson, B.C. (2007). What Do Mentoring and Training in the Responsible Conduct of Research Have To Do with Scientists’ Misbehavior? Findings from a National Survey of NIH-Funded Scientists. Academic Medicine, 82 (9): 853-860.
- Bulger, R.E. & Heitman, E. (2007). Expanding Responsible Conduct of Research Instruction across the University. Academic Medicine, 82 (9): 876-878.
- Kalichman, M.W. (2006). Ethics and Science: A 0.1% solution. Issues in Science and Technology, 23 : 34-36.
- Kalichman, M.W. (2007). Responding to Challenges in Educating for the Responsible Conduct of Research. Academic Medicine, 82 (9): 870-875.
- Kalichman, M.W., Plemmons, D.K. (2007). Reported Goals for Responsible Conduct of Research Courses. Academic Medicine, 82 (9): 846-852.
- Kalichman, M.W. (2009). Evidence-based research ethics. The American Journal of Bioethics, 9 (6&7): 85-87.
- Pimple, K.D. (2002). Six Domains of Research Ethics: A Heuristic Framework for the Responsible Conduct of Research. Science and Engineering Ethics, 8 (2): 191-205.
- Steneck, N.H. (2006). Fostering Integrity in Research: Definitions, Current Knowledge, and Future Directions. Science and Engineering Ethics, 12 : 53-74.
- Steneck, N.H., Bulger, R.E. (2007). The History, Purpose, and Future of Instruction in the Responsible Conduct of Research. Academic Medicine, 82 (9): 829-834.
- Vasgird, D.R. (2007). Prevention over Cure: The Administrative Rationale for Education in the Responsible Conduct of Research. Academic Medicine, 82 (9): 835-837.
- Aristotle. The Nichomachean Ethics .
- Beauchamp, R.L. & Childress, J.F. (2001). Principles of Biomedical Ethics , 5th edition. NY: Oxford University Press.
- Bentham, J. (1781). An Introduction to the Principles of Morals and Legislation.
- Gilligan, C. (1993). In a Different Voice: Psychological Theory and Women’s Development. Cambridge: Harvard University Press.
- Glover, Jonathan. (1977). Causing Death and Saving Lives. Penguin Books.
- Honderich, T, ed. (1995). The Oxford Companion to Philosophy. Oxford and New York: Oxford University Press.
- Kagan, S. (1998). Normative Ethics . Westview Press.
- Kant, I. (1785). Groundwork of the Metaphysics of Morals .
- Kant, I. (1788). Critique of Practical Reason .
- Kant, I. (1797). The Metaphysics of Morals .
- Kant, I. (1797). On a Supposed right to Lie from Benevolent Motives .
- Kuhse, H. & Singer, P. (1999). Bioethics: An Anthology . Blackwell Publishers.
- Mill, J.S. (1861). Utilitarianism.
- Rachels, J. (1999). The Elements of Moral Philosophy , 3rd edition. Boston: McGraw-Hill.
- Regan, T. (1993). Matters of Life and Death: New Introductory Essays in Moral Philosophy , 3rd edition. New York: McGraw-Hill. The history of ethics
- Singer, P (1993). Practical Ethics , 2nd ed. Cambridge University Press
The Importance of Ethics in Research Essay
- To find inspiration for your paper and overcome writer’s block
- As a source of information (ensure proper referencing)
- As a template for you assignment
Introduction
Factors that increase vulnerability of subjects to research abuse, the solution to reduce vulnerability of subjects to research abuse.
In science and medical research, ethics is essential in enhancing the safety and well-being of the subjects or participants. Different studies globally expose vulnerable populations or subjects to abuse, affecting their overall health. In the same case, researchers are employing diverse strategies to enhance ethics and reduce subjects’ vulnerabilities to negative implications of studies and abuse. For these reasons, it is essential to examine the factors that enhance subjects’ vulnerability to abuse and maltreatment during scientific studies. These reasons are economic and financial problems, impractical hope, improper patient advocacy, as well as non-compliance to research ethics. Contrarily, encouraging compliance with ethical principles during research would reduce subjects’ susceptibility to abuse and negative research implications.
Economic and financial issues are among the factors that increase subjects’ vulnerability to abuse during research. Kelly (2013) indicates that “people struggling to put food on the table and a roof over their heads” are vulnerable to abuse during clinical trials and pharmaceutical studies. Washington (2008) also notes that “jobless white men turned their noses at the disgusting work and partly pay” when referring to a perilous clinical experiment conducted in 1935 by the University of Pennsylvania. The sentiment indicates that the university conducted unethical and dangerous medical research on subjects with economic and financial problems (Washington, 2008). The phrase “when you see an opportunity to feed your starving family, […], or get treatment for a terminal disease?” also indicates how financial issues make subjects partake in unsafe clinical trials (Kelly, 2013). Thus, these studies increase the health risks, burden, disparity, and complications among subjects from developing and low-income countries as well as communities.
Unrealistic hope is another factor that increases the risks of abuse among subjects of research activities. According to Washington (2008), patients desperate for healing from diseases, surviving, living longer, and stressed about their health conditions are vulnerable to abuse during medical studies. The phrase “was described to him as his last chance at a meaningful life” shows that James Quinn was a victim of research abuse because of hoped to live a productive life after artificial heart implantation (Washington, 2008). The quote, “Do you have a choice about participating when you see an opportunity to (…) get treatment for a terminal disease,” indicates that subjects are always hopeful of improving their health after clinical studies (Kelly, 2008). This makes patients accept risky treatments or clinical trials that are mentally or physically abusive to their health, hoping to enhance their lifespan.
Improper patient advocacy and education are also major factors that increase the dangers of abusing subjects during clinical research. Washington (2008) questions how medical researchers and providers empower patients about the risks and benefits of clinical trials before treatments. Washington (2008) asks, “But are such warnings offered in a fair and intelligible manner?” The quote proves that researchers obtain patients’ consent for treatments when they are mentally, emotionally, and cognitively incapable of making informed decisions about these interventions. Kelly (2013) also agrees that healthcare researchers fail to provide accurate and quality patient information before clinical trials. The sentence “one of the most commonly cited ethical qualms with clinical trials tends to be misinformation” indicates that patients’ advocacy teams misinform subjects before clinical trials (Kelly, 2013). The wrong information affects the ability of patients to make informed decisions about participating or not partaking in medical studies.
Finally, non-compliance to research ethics and regulations among researchers also makes subjects vulnerable to abuse during studies. Washington (2008) indicates that “the informed consent process consists of much more than obtaining a patient signature on a piece of paper.” The quote implies that medical researchers are violating the informed consent ethics of research that requires patients’ participation only after knowing all the risks and benefits of a treatment. In the statement, “one of the most commonly cited ethical qualms with clinical trials tends to be misinformation,” Kelly (2013) supports Washington (2008) about the voluntary and involuntary deception of subjects to participate in their studies. Generally, violation of ethics makes vulnerable subjects partake in medical or scientific studies that harm their health and those around them.
To reduce the susceptibility of patients or subjects to abusive medical and scientific studies, adherence or compliance to research ethics is essential. For instance, Kelly (2013) suggests that continuous investigation by ethics boards on researchers violating ethical requirements would enhance compliance with research regulations, integrity, and morals. The statement “informed consent is an ongoing process of patient notification and education” also implies that continuous patient advocacy, edification, and communication before treatments and clinical trials is essential in reducing non-compliance to research ethics (Washington, 2008). Moreover, when seeing patient permission for research inclusion, Washington (2008) proposes that researchers should make patients aware of treatment risks through the phrase “the scientist must […] knows all the known risks and must inform the subject proxy.” This will make subjects decide to participate in research after knowing the benefits and negative implications on them and the people around them.
Conclusively, the lack of compliance with ethical research principles among scientists and the economic issues of patients make them susceptible to abuse during studies. Unrealistic hope, desperation, and inappropriate patient advocacy and education make subjects vulnerable to research abuse. Based on these, developing, implementing, and complying with research ethics is a feasible approach to reducing the vulnerability of research subjects to abuse. Therefore, patient abuse is a systemic issue in medical and scientific studies. This means that this problem requires universal or systemic solutions to achieve the desired outcome of protecting patients’ interests and well-being during and after research.
Kelly, S. (2013). Testing drugs on the developing world . The Atlantic. Web.
Washington, H. (2008). Medical apartheid: The dark history of medical experimentation on Black Americans from colonial times to the present. Psychiatric Services , 58 (10), 1380-1381.
- Animal Fur Clothing: The Ethical Issues
- Whistle-Blowing and Ethics: Facebook's Data Selling
- Employment Non-Compliance in Australia in 2008-12
- Donnie Darko by Richard Kelly Review
- Feldman and Kelly's Views on Disagreement
- Practice-Informed Research: Case Analysis
- Ethical Issues Associated with Online Research
- Ethical Principles in Research
- Discussion: Ethics and Fashion
- Abortion Rights: The Ethical Issues
- Chicago (A-D)
- Chicago (N-B)
IvyPanda. (2024, April 1). The Importance of Ethics in Research. https://ivypanda.com/essays/the-importance-of-ethics-in-research/
"The Importance of Ethics in Research." IvyPanda , 1 Apr. 2024, ivypanda.com/essays/the-importance-of-ethics-in-research/.
IvyPanda . (2024) 'The Importance of Ethics in Research'. 1 April.
IvyPanda . 2024. "The Importance of Ethics in Research." April 1, 2024. https://ivypanda.com/essays/the-importance-of-ethics-in-research/.
1. IvyPanda . "The Importance of Ethics in Research." April 1, 2024. https://ivypanda.com/essays/the-importance-of-ethics-in-research/.
Bibliography
IvyPanda . "The Importance of Ethics in Research." April 1, 2024. https://ivypanda.com/essays/the-importance-of-ethics-in-research/.
IvyPanda uses cookies and similar technologies to enhance your experience, enabling functionalities such as:
- Basic site functions
- Ensuring secure, safe transactions
- Secure account login
- Remembering account, browser, and regional preferences
- Remembering privacy and security settings
- Analyzing site traffic and usage
- Personalized search, content, and recommendations
- Displaying relevant, targeted ads on and off IvyPanda
Please refer to IvyPanda's Cookies Policy and Privacy Policy for detailed information.
Certain technologies we use are essential for critical functions such as security and site integrity, account authentication, security and privacy preferences, internal site usage and maintenance data, and ensuring the site operates correctly for browsing and transactions.
Cookies and similar technologies are used to enhance your experience by:
- Remembering general and regional preferences
- Personalizing content, search, recommendations, and offers
Some functions, such as personalized recommendations, account preferences, or localization, may not work correctly without these technologies. For more details, please refer to IvyPanda's Cookies Policy .
To enable personalized advertising (such as interest-based ads), we may share your data with our marketing and advertising partners using cookies and other technologies. These partners may have their own information collected about you. Turning off the personalized advertising setting won't stop you from seeing IvyPanda ads, but it may make the ads you see less relevant or more repetitive.
Personalized advertising may be considered a "sale" or "sharing" of the information under California and other state privacy laws, and you may have the right to opt out. Turning off personalized advertising allows you to exercise your right to opt out. Learn more in IvyPanda's Cookies Policy and Privacy Policy .

What is Ethics in Research and Why is it Important?
This article defines ethics in research, including codes and policies, and demonstrates the importance of ethics in research.
- Privacy Policy

Home » Ethical Considerations – Types, Examples and Writing Guide
Ethical Considerations – Types, Examples and Writing Guide
Table of Contents

Ethical Considerations
Ethical considerations in research refer to the principles and guidelines that researchers must follow to ensure that their studies are conducted in an ethical and responsible manner. These considerations are designed to protect the rights, safety, and well-being of research participants, as well as the integrity and credibility of the research itself
Some of the key ethical considerations in research include:
- Informed consent: Researchers must obtain informed consent from study participants, which means they must inform participants about the study’s purpose, procedures, risks, benefits, and their right to withdraw at any time.
- Privacy and confidentiality : Researchers must ensure that participants’ privacy and confidentiality are protected. This means that personal information should be kept confidential and not shared without the participant’s consent.
- Harm reduction : Researchers must ensure that the study does not harm the participants physically or psychologically. They must take steps to minimize the risks associated with the study.
- Fairness and equity : Researchers must ensure that the study does not discriminate against any particular group or individual. They should treat all participants equally and fairly.
- Use of deception: Researchers must use deception only if it is necessary to achieve the study’s objectives. They must inform participants of the deception as soon as possible.
- Use of vulnerable populations : Researchers must be especially cautious when working with vulnerable populations, such as children, pregnant women, prisoners, and individuals with cognitive or intellectual disabilities.
- Conflict of interest : Researchers must disclose any potential conflicts of interest that may affect the study’s integrity. This includes financial or personal relationships that could influence the study’s results.
- Data manipulation: Researchers must not manipulate data to support a particular hypothesis or agenda. They should report the results of the study objectively, even if the findings are not consistent with their expectations.
- Intellectual property: Researchers must respect intellectual property rights and give credit to previous studies and research.
- Cultural sensitivity : Researchers must be sensitive to the cultural norms and beliefs of the participants. They should avoid imposing their values and beliefs on the participants and should be respectful of their cultural practices.
Types of Ethical Considerations
Types of Ethical Considerations are as follows:
Research Ethics:
This includes ethical principles and guidelines that govern research involving human or animal subjects, ensuring that the research is conducted in an ethical and responsible manner.
Business Ethics :
This refers to ethical principles and standards that guide business practices and decision-making, such as transparency, honesty, fairness, and social responsibility.
Medical Ethics :
This refers to ethical principles and standards that govern the practice of medicine, including the duty to protect patient autonomy, informed consent, confidentiality, and non-maleficence.
Environmental Ethics :
This involves ethical principles and values that guide our interactions with the natural world, including the obligation to protect the environment, minimize harm, and promote sustainability.
Legal Ethics
This involves ethical principles and standards that guide the conduct of legal professionals, including issues such as confidentiality, conflicts of interest, and professional competence.
Social Ethics
This involves ethical principles and values that guide our interactions with other individuals and society as a whole, including issues such as justice, fairness, and human rights.
Information Ethics
This involves ethical principles and values that govern the use and dissemination of information, including issues such as privacy, accuracy, and intellectual property.
Cultural Ethics
This involves ethical principles and values that govern the relationship between different cultures and communities, including issues such as respect for diversity, cultural sensitivity, and inclusivity.
Technological Ethics
This refers to ethical principles and guidelines that govern the development, use, and impact of technology, including issues such as privacy, security, and social responsibility.
Journalism Ethics
This involves ethical principles and standards that guide the practice of journalism, including issues such as accuracy, fairness, and the public interest.
Educational Ethics
This refers to ethical principles and standards that guide the practice of education, including issues such as academic integrity, fairness, and respect for diversity.
Political Ethics
This involves ethical principles and values that guide political decision-making and behavior, including issues such as accountability, transparency, and the protection of civil liberties.
Professional Ethics
This refers to ethical principles and standards that guide the conduct of professionals in various fields, including issues such as honesty, integrity, and competence.
Personal Ethics
This involves ethical principles and values that guide individual behavior and decision-making, including issues such as personal responsibility, honesty, and respect for others.
Global Ethics
This involves ethical principles and values that guide our interactions with other nations and the global community, including issues such as human rights, environmental protection, and social justice.
Applications of Ethical Considerations
Ethical considerations are important in many areas of society, including medicine, business, law, and technology. Here are some specific applications of ethical considerations:
- Medical research : Ethical considerations are crucial in medical research, particularly when human subjects are involved. Researchers must ensure that their studies are conducted in a way that does not harm participants and that participants give informed consent before participating.
- Business practices: Ethical considerations are also important in business, where companies must make decisions that are socially responsible and avoid activities that are harmful to society. For example, companies must ensure that their products are safe for consumers and that they do not engage in exploitative labor practices.
- Environmental protection: Ethical considerations play a crucial role in environmental protection, as companies and governments must weigh the benefits of economic development against the potential harm to the environment. Decisions about land use, resource allocation, and pollution must be made in an ethical manner that takes into account the long-term consequences for the planet and future generations.
- Technology development : As technology continues to advance rapidly, ethical considerations become increasingly important in areas such as artificial intelligence, robotics, and genetic engineering. Developers must ensure that their creations do not harm humans or the environment and that they are developed in a way that is fair and equitable.
- Legal system : The legal system relies on ethical considerations to ensure that justice is served and that individuals are treated fairly. Lawyers and judges must abide by ethical standards to maintain the integrity of the legal system and to protect the rights of all individuals involved.
Examples of Ethical Considerations
Here are a few examples of ethical considerations in different contexts:
- In healthcare : A doctor must ensure that they provide the best possible care to their patients and avoid causing them harm. They must respect the autonomy of their patients, and obtain informed consent before administering any treatment or procedure. They must also ensure that they maintain patient confidentiality and avoid any conflicts of interest.
- In the workplace: An employer must ensure that they treat their employees fairly and with respect, provide them with a safe working environment, and pay them a fair wage. They must also avoid any discrimination based on race, gender, religion, or any other characteristic protected by law.
- In the media : Journalists must ensure that they report the news accurately and without bias. They must respect the privacy of individuals and avoid causing harm or distress. They must also be transparent about their sources and avoid any conflicts of interest.
- In research: Researchers must ensure that they conduct their studies ethically and with integrity. They must obtain informed consent from participants, protect their privacy, and avoid any harm or discomfort. They must also ensure that their findings are reported accurately and without bias.
- In personal relationships : People must ensure that they treat others with respect and kindness, and avoid causing harm or distress. They must respect the autonomy of others and avoid any actions that would be considered unethical, such as lying or cheating. They must also respect the confidentiality of others and maintain their privacy.
How to Write Ethical Considerations
When writing about research involving human subjects or animals, it is essential to include ethical considerations to ensure that the study is conducted in a manner that is morally responsible and in accordance with professional standards. Here are some steps to help you write ethical considerations:
- Describe the ethical principles: Start by explaining the ethical principles that will guide the research. These could include principles such as respect for persons, beneficence, and justice.
- Discuss informed consent : Informed consent is a critical ethical consideration when conducting research. Explain how you will obtain informed consent from participants, including how you will explain the purpose of the study, potential risks and benefits, and how you will protect their privacy.
- Address confidentiality : Describe how you will protect the confidentiality of the participants’ personal information and data, including any measures you will take to ensure that the data is kept secure and confidential.
- Consider potential risks and benefits : Describe any potential risks or harms to participants that could result from the study and how you will minimize those risks. Also, discuss the potential benefits of the study, both to the participants and to society.
- Discuss the use of animals : If the research involves the use of animals, address the ethical considerations related to animal welfare. Explain how you will minimize any potential harm to the animals and ensure that they are treated ethically.
- Mention the ethical approval : Finally, it’s essential to acknowledge that the research has received ethical approval from the relevant institutional review board or ethics committee. State the name of the committee, the date of approval, and any specific conditions or requirements that were imposed.
When to Write Ethical Considerations
Ethical considerations should be written whenever research involves human subjects or has the potential to impact human beings, animals, or the environment in some way. Ethical considerations are also important when research involves sensitive topics, such as mental health, sexuality, or religion.
In general, ethical considerations should be an integral part of any research project, regardless of the field or subject matter. This means that they should be considered at every stage of the research process, from the initial planning and design phase to data collection, analysis, and dissemination.
Ethical considerations should also be written in accordance with the guidelines and standards set by the relevant regulatory bodies and professional associations. These guidelines may vary depending on the discipline, so it is important to be familiar with the specific requirements of your field.
Purpose of Ethical Considerations
Ethical considerations are an essential aspect of many areas of life, including business, healthcare, research, and social interactions. The primary purposes of ethical considerations are:
- Protection of human rights: Ethical considerations help ensure that people’s rights are respected and protected. This includes respecting their autonomy, ensuring their privacy is respected, and ensuring that they are not subjected to harm or exploitation.
- Promoting fairness and justice: Ethical considerations help ensure that people are treated fairly and justly, without discrimination or bias. This includes ensuring that everyone has equal access to resources and opportunities, and that decisions are made based on merit rather than personal biases or prejudices.
- Promoting honesty and transparency : Ethical considerations help ensure that people are truthful and transparent in their actions and decisions. This includes being open and honest about conflicts of interest, disclosing potential risks, and communicating clearly with others.
- Maintaining public trust: Ethical considerations help maintain public trust in institutions and individuals. This is important for building and maintaining relationships with customers, patients, colleagues, and other stakeholders.
- Ensuring responsible conduct: Ethical considerations help ensure that people act responsibly and are accountable for their actions. This includes adhering to professional standards and codes of conduct, following laws and regulations, and avoiding behaviors that could harm others or damage the environment.
Advantages of Ethical Considerations
Here are some of the advantages of ethical considerations:
- Builds Trust : When individuals or organizations follow ethical considerations, it creates a sense of trust among stakeholders, including customers, clients, and employees. This trust can lead to stronger relationships and long-term loyalty.
- Reputation and Brand Image : Ethical considerations are often linked to a company’s brand image and reputation. By following ethical practices, a company can establish a positive image and reputation that can enhance its brand value.
- Avoids Legal Issues: Ethical considerations can help individuals and organizations avoid legal issues and penalties. By adhering to ethical principles, companies can reduce the risk of facing lawsuits, regulatory investigations, and fines.
- Increases Employee Retention and Motivation: Employees tend to be more satisfied and motivated when they work for an organization that values ethics. Companies that prioritize ethical considerations tend to have higher employee retention rates, leading to lower recruitment costs.
- Enhances Decision-making: Ethical considerations help individuals and organizations make better decisions. By considering the ethical implications of their actions, decision-makers can evaluate the potential consequences and choose the best course of action.
- Positive Impact on Society: Ethical considerations have a positive impact on society as a whole. By following ethical practices, companies can contribute to social and environmental causes, leading to a more sustainable and equitable society.
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like

Assignment – Types, Examples and Writing Guide

Research Problem – Examples, Types and Guide

Research Paper – Structure, Examples and Writing...

Figures in Research Paper – Examples and Guide

Data Interpretation – Process, Methods and...

Institutional Review Board – Application Sample...
Ethical considerations in research: Best practices and examples

To conduct responsible research, you’ve got to think about ethics. They protect participants’ rights and their well-being - and they ensure your findings are valid and reliable. This isn’t just a box for you to tick. It’s a crucial consideration that can make all the difference to the outcome of your research.
In this article, we'll explore the meaning and importance of research ethics in today's research landscape. You'll learn best practices to conduct ethical and impactful research.
Examples of ethical considerations in research
As a researcher, you're responsible for ethical research alongside your organization. Fulfilling ethical guidelines is critical. Organizations must ensure employees follow best practices to protect participants' rights and well-being.
Keep these things in mind when it comes to ethical considerations in research:
Voluntary participation
Voluntary participation is key. Nobody should feel like they're being forced to participate or pressured into doing anything they don't want to. That means giving people a choice and the ability to opt out at any time, even if they've already agreed to take part in the study.
Informed consent
Informed consent isn't just an ethical consideration. It's a legal requirement as well. Participants must fully understand what they're agreeing to, including potential risks and benefits.
The best way to go about this is by using a consent form. Make sure you include:
- A brief description of the study and research methods.
- The potential benefits and risks of participating.
- The length of the study.
- Contact information for the researcher and/or sponsor.
- Reiteration of the participant’s right to withdraw from the research project at any time without penalty.
Anonymity means that participants aren't identifiable in any way. This includes:
- Email address
- Photographs
- Video footage
You need a way to anonymize research data so that it can't be traced back to individual participants. This may involve creating a new digital ID for participants that can’t be linked back to their original identity using numerical codes.
Confidentiality
Information gathered during a study must be kept confidential. Confidentiality helps to protect the privacy of research participants. It also ensures that their information isn't disclosed to unauthorized individuals.
Some ways to ensure confidentiality include:
- Using a secure server to store data.
- Removing identifying information from databases that contain sensitive data.
- Using a third-party company to process and manage research participant data.
- Not keeping participant records for longer than necessary.
- Avoiding discussion of research findings in public forums.
Potential for harm
The potential for harm is a crucial factor in deciding whether a research study should proceed. It can manifest in various forms, such as:
- Psychological harm
- Social harm
- Physical harm
Conduct an ethical review to identify possible harms. Be prepared to explain how you’ll minimize these harms and what support is available in case they do happen.
Fair payment
One of the most crucial aspects of setting up a research study is deciding on fair compensation for your participants. Underpayment is a common ethical issue that shouldn't be overlooked. Properly rewarding participants' time is critical for boosting engagement and obtaining high-quality data. While Prolific requires a minimum payment of £6.00 / $8.00 per hour, there are other factors you need to consider when deciding on a fair payment.
First, check your institution's reimbursement guidelines to see if they already have a minimum or maximum hourly rate. You can also use the national minimum wage as a reference point.
Next, think about the amount of work you're asking participants to do. The level of effort required for a task, such as producing a video recording versus a short survey, should correspond with the reward offered.
You also need to consider the population you're targeting. To attract research subjects with specific characteristics or high-paying jobs, you may need to offer more as an incentive.
We recommend a minimum payment of £9.00 / $12.00 per hour, but we understand that payment rates can vary depending on a range of factors. Whatever payment you choose should reflect the amount of effort participants are required to put in and be fair to everyone involved.
Ethical research made easy with Prolific
At Prolific, we believe in making ethical research easy and accessible. The findings from the Fairwork Cloudwork report speak for themselves. Prolific was given the top score out of all competitors for minimum standards of fair work.
With over 25,000 researchers in our community, we're leading the way in revolutionizing the research industry. If you're interested in learning more about how we can support your research journey, sign up to get started now.
You might also like

High-quality human data to deliver world-leading research and AIs.

Follow us on
All Rights Reserved Prolific 2024

Understanding ethics approval in academic research
The following strategies aim to streamline the ethics approval process, reduce delays and uphold the highest ethical standards
Marios Kremantzis
.css-76pyzs{margin-right:0.25rem;} ,, fatema zaghloul, rushana khusainova.

Created in partnership with

You may also like
Popular resources
.css-1txxx8u{overflow:hidden;max-height:81px;text-indent:0px;} Change is coming, whether higher education likes it or not
Is it worth paying for genai, emotions and learning: what role do emotions play in how and why students learn, teaching international students about academic integrity, ai and assessment redesign: a four-step process.
Ethical considerations are foundational to the integrity and validity of scholarly work, guiding researchers to conduct studies that are academically sound and morally responsible. Securing ethics approval is critical for group or individual research projects at any academic level. This process ensures the research respects and protects participants' rights, dignity and welfare.
However, students and supervisors often face challenges that can delay or obstruct approval, such as a lack of understanding of ethical guidelines and inadequate preparation of ethics applications. Students might struggle with articulating study risks and benefits or ensuring data confidentiality, especially in research involving vulnerable populations or sensitive topics. Supervisors may find it difficult to guide students through ethical research practices while balancing academic rigour and ethical responsibility .
Common problems in ethics applications
Documentation and consent issues: the omission of crucial documents (eg, participant information sheets, consent forms, questionnaires or interview guides) is a common oversight. These documents are crucial for informing participants about the research and securing their informed consent.
“Respect for persons” entails acknowledging the autonomy of all participants, obtaining informed consent and ensuring that they are fully aware of their rights to withdraw from the study at any point.
Privacy and security concerns: failing to mention which online tools will be used for video interviews can lead to ethical and technical issues. It is essential to specify platforms to ensure they meet privacy and security standards. When researchers do not disclose the specific platforms they intend to use, they risk compromising the confidentiality and security of participant data.
Ethical issues arise when participants are not adequately informed about how their data will be collected, stored and protected. This lack of transparency can undermine the trust between researchers and participants, potentially leading to a reluctance to participate or the withdrawal of consent.
Project clarity and methodological details: despite the fact that ambiguity can result in misunderstandings about the research aims, methods and potential impacts, applications often lack a clear description of the project. Without this, ethics review boards cannot accurately assess the ethical implications of the research.
Applications frequently lack details about methodological choices, such as research instruments, data collection processes and sampling strategies. Clear, detailed methodologies are necessary to evaluate the ethical implications fully.
Power dynamics and confidentiality: some students overlook power dynamics and gatekeeping issues. For example, recruiting staff members through line managers can introduce significant power imbalances. Ensuring anonymity and confidentiality is also often inadequately addressed.
Software and data analysis: the lack of specifics regarding software tools for data analysis and storage can be problematic. Researchers must provide details about the tools to be used to ensure they comply with ethical standards.
Primary and secondary data use: there is often a lack of understanding regarding the differences between primary and secondary data. Some students fail to specify the databases they intend to use or attempt to use non-subscribed databases.
Supervisor review and endorsement: there have been instances where forms lack the supervisor’s signature. This indicates that the application has not been thoroughly reviewed and endorsed by a faculty member.
Data storage and security: students sometimes fail to mention how they will store their primary data securely, which is essential for protecting participant confidentiality. Secure data storage practices are vital for preventing unauthorised access and ensuring that data is preserved accurately and ethically.
- Resource collection: Research excellence: what is it and how can universities achieve it?
- How to add value to research and manage intellectual property
- A model for maximising the impact of small research units
Strategies for overcoming obstacles
To address these common problems, we can implement several effective strategies:
Developing and distributing standardised checklists: supervisors can use these when reviewing student applications to ensure all essential components are included and identify missing elements quickly. These checklists should cover participant information, consent forms, specified tools for data collection and data storage plans.
Integrating checklists into the university’s research ethics management system: this would streamline the review process, enabling supervisors to ensure all criteria are met before approving applications. By configuring the system to recognise primary data collection, it can mandate attaching relevant files (eg, recruitment materials and participant information sheets).
Creating a centralised repository of resources: this should include exemplary ethics applications submitted and approved in the past, which would provide supervisors and students with clear examples of best practices.
Offering training sessions and workshops on ethical research practices: this helps students and supervisors understand and address ethical considerations in their applications. The dissertation handbook should include detailed guidelines on ethical research. Consistent online training should be provided for staff and students before and during the initial months of the dissertation process. These sessions should cover topics such as informed consent, confidentiality and managing sensitive data. Additionally, incorporating case studies and real-life scenarios in these workshops provides practical insights into handling ethical dilemmas effectively.
Providing clear guidelines on the use of software tools and data sources: including a list of approved databases and platforms can prevent confusion and ensure compliance with institutional policies.
Addressing ethics application issues proactively streamlines the approval process, enhances research quality and upholds ethical standards. Informing and preparing students and supervisors fosters an ethical research environment, while comprehensive resources empower researchers to navigate challenges responsibly and with integrity.
Marios Kremantzis and Fatema Zaghloul are lecturers in business analytics and Rushana Khusainova is a senior lecturer in marketing at the University of Bristol Business School.
If you would like advice and insight from academics and university staff delivered direct to your inbox each week, sign up for the Campus newsletter .
The iScanner app supports the academic community in information sharing and management
Change is coming, whether higher education likes it or not, use ai to get your students thinking critically, a diy guide to starting your own journal, artificial intelligence and academic integrity: striking a balance, contextual learning: linking learning to the real world.
Register for free
and unlock a host of features on the THE site
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Springer Nature - PMC COVID-19 Collection

Ethical Issues in Research: Perceptions of Researchers, Research Ethics Board Members and Research Ethics Experts
Marie-josée drolet.
1 Department of Occupational Therapy (OT), Université du Québec à Trois-Rivières (UQTR), Trois-Rivières (Québec), Canada
Eugénie Rose-Derouin
2 Bachelor OT program, Université du Québec à Trois-Rivières (UQTR), Trois-Rivières (Québec), Canada
Julie-Claude Leblanc
Mélanie ruest, bryn williams-jones.
3 Department of Social and Preventive Medicine, School of Public Health, Université de Montréal, Montréal (Québec), Canada
In the context of academic research, a diversity of ethical issues, conditioned by the different roles of members within these institutions, arise. Previous studies on this topic addressed mainly the perceptions of researchers. However, to our knowledge, no studies have explored the transversal ethical issues from a wider spectrum, including other members of academic institutions as the research ethics board (REB) members, and the research ethics experts. The present study used a descriptive phenomenological approach to document the ethical issues experienced by a heterogeneous group of Canadian researchers, REB members, and research ethics experts. Data collection involved socio-demographic questionnaires and individual semi-structured interviews. Following the triangulation of different perspectives (researchers, REB members and ethics experts), emerging ethical issues were synthesized in ten units of meaning: (1) research integrity, (2) conflicts of interest, (3) respect for research participants, (4) lack of supervision and power imbalances, (5) individualism and performance, (6) inadequate ethical guidance, (7) social injustices, (8) distributive injustices, (9) epistemic injustices, and (10) ethical distress. This study highlighted several problematic elements that can support the identification of future solutions to resolve transversal ethical issues in research that affect the heterogeneous members of the academic community.
Introduction
Research includes a set of activities in which researchers use various structured methods to contribute to the development of knowledge, whether this knowledge is theoretical, fundamental, or applied (Drolet & Ruest, accepted ). University research is carried out in a highly competitive environment that is characterized by ever-increasing demands (i.e., on time, productivity), insufficient access to research funds, and within a market economy that values productivity and speed often to the detriment of quality or rigour – this research context creates a perfect recipe for breaches in research ethics, like research misbehaviour or misconduct (i.e., conduct that is ethically questionable or unacceptable because it contravenes the accepted norms of responsible conduct of research or compromises the respect of core ethical values that are widely held by the research community) (Drolet & Girard, 2020 ; Sieber, 2004 ). Problematic ethics and integrity issues – e.g., conflicts of interest, falsification of data, non-respect of participants’ rights, and plagiarism, to name but a few – have the potential to both undermine the credibility of research and lead to negative consequences for many stakeholders, including researchers, research assistants and personnel, research participants, academic institutions, and society as a whole (Drolet & Girard, 2020 ). It is thus evident that the academic community should be able to identify these different ethical issues in order to evaluate the nature of the risks that they pose (and for whom), and then work towards their prevention or management (i.e., education, enhanced policies and procedures, risk mitigation strategies).
In this article, we define an “ethical issue” as any situation that may compromise, in whole or in part, the respect of at least one moral value (Swisher et al., 2005 ) that is considered socially legitimate and should thus be respected. In general, ethical issues occur at three key moments or stages of the research process: (1) research design (i.e., conception, project planning), (2) research conduct (i.e., data collection, data analysis) and (3) knowledge translation or communication (e.g., publications of results, conferences, press releases) (Drolet & Ruest, accepted ). According to Sieber ( 2004 ), ethical issues in research can be classified into five categories, related to: (a) communication with participants and the community, (b) acquisition and use of research data, (c) external influence on research, (d) risks and benefits of the research, and (e) selection and use of research theories and methods. Many of these issues are related to breaches of research ethics norms, misbehaviour or research misconduct. Bruhn et al., ( 2002 ) developed a typology of misbehaviour and misconduct in academia that can be used to judge the seriousness of different cases. This typology takes into consideration two axes of reflection: (a) the origin of the situation (i.e., is it the researcher’s own fault or due to the organizational context?), and (b) the scope and severity (i.e., is this the first instance or a recurrent behaviour? What is the nature of the situation? What are the consequences, for whom, for how many people, and for which organizations?).
A previous detailed review of the international literature on ethical issues in research revealed several interesting findings (Beauchemin et al., 2021 ). Indeed, the current literature is dominated by descriptive ethics, i.e., the sharing by researchers from various disciplines of the ethical issues they have personally experienced. While such anecdotal documentation is relevant, it is insufficient because it does not provide a global view of the situation. Among the reviewed literature, empirical studies were in the minority (Table 1 ) – only about one fifth of the sample (n = 19) presented empirical research findings on ethical issues in research. The first of these studies was conducted almost 50 years ago (Hunt et al., 1984 ), with the remainder conducted in the 1990s. Eight studies were conducted in the United States (n = 8), five in Canada (n = 5), three in England (n = 3), two in Sweden (n = 2) and one in Ghana (n = 1).
Summary of Empirical Studies on Ethical Issues in Research by the year of publication
| References | Country | Types of research participants | Study design |
|---|---|---|---|
| Hunt et al., ( ) | USA | marketing researchers | mixed-methods |
| Pope & Vetter ( ) | USA | members of the American psychological association | quantitative |
| Swazey et al., ( ) | USA | doctoral candidates and faculty members | quantitative |
| Balk ( ) | USA | study participants | mixed-methods |
| Sigmon ( ) | USA | psychopathology researchers | quantitative |
| Fraser ( ) | UK | education researchers | qualitative |
| Lynöe et al., ( ) | Sweden | research ethics board members, researchers, healthcare politicians and district nurses | quantitative |
| Bouffard ( ) | Canada | researchers, health professionals and patients | qualitative |
| Davison ( ) | UK | social work researchers | qualitative |
| Miyazaki & Taylor ( ) | USA | non-traditional undergraduate students | quantitative |
| Mondain & Bologo ( ) | Ghana | researcher participants and other stakeholders | qualitative |
| Wiegand & Funk ( ) | Canada | nurses | quantitative |
| McGinn ( ) | USA | nanotechnology researchers | quantitative |
| Colnerud ( ) | Sweden | researchers | qualitative |
| Lierville et al., ( ) | Canada | Managers, Researchers, Unit Leaders and Practitioners | Qualitative |
| Giorgini et al., ( ) | USA | researchers | mixed-methods |
| Birchley et al., ( ) | UK | smart-home researchers | qualitative |
| Jarvis ( ) | Canada | research participants (women and their family members), health care providers and key stakeholders | qualitative |
| Drolet & Girard ( ) | Canada | occupational therapist researchers | qualitative |
Further, the majority of studies in our sample (n = 12) collected the perceptions of a homogeneous group of participants, usually researchers (n = 14) and sometimes health professionals (n = 6). A minority of studies (n = 7) triangulated the perceptions of diverse research stakeholders (i.e., researchers and research participants, or students). To our knowledge, only one study has examined perceptions of ethical issues in research by research ethics board members (REB; Institutional Review Boards [IRB] in the USA), and none to date have documented the perceptions of research ethics experts. Finally, nine studies (n = 9) adopted a qualitative design, seven studies (n = 7) a quantitative design, and three (n = 3) a mixed-methods design.
More studies using empirical research methods are needed to better identify broader trends, to enrich discussions on the values that should govern responsible conduct of research in the academic community, and to evaluate the means by which these values can be supported in practice (Bahn, 2012 ; Beauchemin et al., 2021 ; Bruhn et al., 2002 ; Henderson et al., 2013 ; Resnik & Elliot, 2016; Sieber 2004 ). To this end, we conducted an empirical qualitative study to document the perceptions and experiences of a heterogeneous group of Canadian researchers, REB members, and research ethics experts, to answer the following broad question: What are the ethical issues in research?
Research Methods
Research design.
A qualitative research approach involving individual semi-structured interviews was used to systematically document ethical issues (De Poy & Gitlin, 2010 ; Hammell et al., 2000 ). Specifically, a descriptive phenomenological approach inspired by the philosophy of Husserl was used (Husserl, 1970 , 1999 ), as it is recommended for documenting the perceptions of ethical issues raised by various practices (Hunt & Carnavale, 2011 ).
Ethical considerations
The principal investigator obtained ethics approval for this project from the Research Ethics Board of the Université du Québec à Trois-Rivières (UQTR). All members of the research team signed a confidentiality agreement, and research participants signed the consent form after reading an information letter explaining the nature of the research project.
Sampling and recruitment
As indicated above, three types of participants were sought: (1) researchers from different academic disciplines conducting research (i.e., theoretical, fundamental or empirical) in Canadian universities; (2) REB members working in Canadian organizations responsible for the ethical review, oversight or regulation of research; and (3) research ethics experts, i.e., academics or ethicists who teach research ethics, conduct research in research ethics, or are scholars who have acquired a specialization in research ethics. To be included in the study, participants had to work in Canada, speak and understand English or French, and be willing to participate in the study. Following Thomas and Polio’s (2002) recommendation to recruit between six and twelve participants (for a homogeneous sample) to ensure data saturation, for our heterogeneous sample, we aimed to recruit approximately twelve participants in order to obtain data saturation. Having used this method several times in related projects in professional ethics, data saturation is usually achieved with 10 to 15 participants (Drolet & Goulet, 2018 ; Drolet & Girard, 2020 ; Drolet et al., 2020 ). From experience, larger samples only serve to increase the degree of data saturation, especially in heterogeneous samples (Drolet et al., 2017 , 2019 ; Drolet & Maclure, 2016 ).
Purposive sampling facilitated the identification of participants relevant to documenting the phenomenon in question (Fortin, 2010 ). To ensure a rich and most complete representation of perceptions, we sought participants with varied and complementary characteristics with regards to the social roles they occupy in research practice (Drolet & Girard, 2020 ). A triangulation of sources was used for the recruitment (Bogdan & Biklen, 2006 ). The websites of Canadian universities and Canadian health institution REBs, as well as those of major Canadian granting agencies (i.e., the Canadian Institutes of Health Research, the Natural Sciences and Engineering Research Council of Canada, and the Social Sciences and Humanities Research Council of Canada, Fonds de recherche du Quebec), were searched to identify individuals who might be interested in participating in the study. Further, people known by the research team for their knowledge and sensitivity to ethical issues in research were asked to participate. Research participants were also asked to suggest other individuals who met the study criteria.
Data Collection
Two tools were used for data collecton: (a) a socio-demographic questionnaire, and (b) a semi-structured individual interview guide. English and French versions of these two documents were used and made available, depending on participant preferences. In addition, although the interview guide contained the same questions, they were adapted to participants’ specific roles (i.e., researcher, REB member, research ethics expert). When contacted by email by the research assistant, participants were asked to confirm under which role they wished to participate (because some participants might have multiple, overlapping responsibilities) and they were sent the appropriate interview guide.
The interview guides each had two parts: an introduction and a section on ethical issues. The introduction consisted of general questions to put the participant at ease (i.e., “Tell me what a typical day at work is like for you”). The section on ethical issues was designed to capture the participant’s perceptions through questions such as: “Tell me three stories you have experienced at work that involve an ethical issue?” and “Do you feel that your organization is doing enough to address, manage, and resolve ethical issues in your work?”. Although some interviews were conducted in person, the majority were conducted by videoconference to promote accessibility and because of the COVID-19 pandemic. Interviews were digitally recorded so that the verbatim could be transcribed in full, and varied between 40 and 120 min in duration, with an average of 90 min. Research assistants conducted the interviews and transcribed the verbatim.
Data Analysis
The socio-demographic questionnaires were subjected to simple descriptive statistical analyses (i.e., means and totals), and the semi-structured interviews were subjected to qualitative analysis. The steps proposed by Giorgi ( 1997 ) for a Husserlian phenomenological reduction of the data were used. After collecting, recording, and transcribing the interviews, all verbatim were analyzed by at least two analysts: a research assistant (2nd author of this article) and the principal investigator (1st author) or a postdoctoral fellow (3rd author). The repeated reading of the verbatim allowed the first analyst to write a synopsis, i.e., an initial extraction of units of meaning. The second analyst then read the synopses, which were commented and improved if necessary. Agreement between analysts allowed the final drafting of the interview synopses, which were then analyzed by three analysts to generate and organize the units of meaning that emerged from the qualitative data.
Participants
Sixteen individuals (n = 16) participated in the study, of whom nine (9) identified as female and seven (7) as male (Table 2 ). Participants ranged in age from 22 to 72 years, with a mean age of 47.5 years. Participants had between one (1) and 26 years of experience in the research setting, with an average of 14.3 years of experience. Participants held a variety of roles, including: REB members (n = 11), researchers (n = 10), research ethics experts (n = 4), and research assistant (n = 1). As mentioned previously, seven (7) participants held more than one role, i.e., REB member, research ethics expert, and researcher. The majority (87.5%) of participants were working in Quebec, with the remaining working in other Canadian provinces. Although all participants considered themselves to be francophone, one quarter (n = 4) identified themselves as belonging to a cultural minority group.
Description of Participants
| Participant number | Gender | Age | Year(s) of experience | Participant’s role(s) |
|---|---|---|---|---|
| P1 | F | 20–25 | 1–5 | REB member, and research assistant |
| P2 | F | 45–50 | 10–15 | REB member |
| P3 | F | 35–40 | 20–25 | Researcher |
| P4 | H | 55–60 | 20–25 | REB member, research ethics expert, and researcher |
| P5 | H | 70–75 | 20–25 | REB member and researcher |
| P6 | H | 45–50 | 5–10 | REB member |
| P7 | H | 40–45 | 5–10 | REB member, research ethics expert, and researcher |
| P8 | H | 45–50 | 15–20 | REB member, research ethics expert, and researcher |
| P9 | F | 35–40 | 5–10 | REB member |
| P10 | F | 65–70 | 25–30 | Researcher and research ethics expert |
| P11 | F | 60–65 | 20–25 | REB member |
| P12 | F | 45 − 40 | 20–25 | Researcher |
| P13 | F | 40–45 | 5–10 | REB member |
| P14 | H | 30–35 | 1–15 | Researcher |
| P15 | F | 40–45 | 5–10 | REB member and researcher |
| P16 | H | 50–55 | 20–25 | Researcher |
With respect to their academic background, most participants (n = 9) had a PhD, three (3) had a post-doctorate, two (2) had a master’s degree, and two (2) had a bachelor’s degree. Participants came from a variety of disciplines: nine (9) had a specialty in the humanities or social sciences, four (4) in the health sciences and three (3) in the natural sciences. In terms of their knowledge of ethics, five (5) participants reported having taken one university course entirely dedicated to ethics, four (4) reported having taken several university courses entirely dedicated to ethics, three (3) had a university degree dedicated to ethics, while two (2) only had a few hours or days of training in ethics and two (2) reported having no knowledge of ethics.
Ethical issues
As Fig. 1 illustrates, ten units of meaning emerge from the data analysis, namely: (1) research integrity, (2) conflicts of interest, (3) respect for research participants, (4) lack of supervision and power imbalances, (5) individualism and performance, (6) inadequate ethical guidance, (7) social injustices, (8) distributive injustices, (9) epistemic injustices, and (10) ethical distress. To illustrate the results, excerpts from verbatim interviews are presented in the following sub-sections. Most of the excerpts have been translated into English as the majority of interviews were conducted with French-speaking participants.

Ethical issues in research according to the participants
Research Integrity
The research environment is highly competitive and performance-based. Several participants, in particular researchers and research ethics experts, felt that this environment can lead both researchers and research teams to engage in unethical behaviour that reflects a lack of research integrity. For example, as some participants indicated, competition for grants and scientific publications is sometimes so intense that researchers falsify research results or plagiarize from colleagues to achieve their goals.
Some people will lie or exaggerate their research findings in order to get funding. Then, you see it afterwards, you realize: “ah well, it didn’t work, but they exaggerated what they found and what they did” (participant 14). Another problem in research is the identification of authors when there is a publication. Very often, there are authors who don’t even know what the publication is about and that their name is on it. (…) The time that it surprised me the most was just a few months ago when I saw someone I knew who applied for a teaching position. He got it I was super happy for him. Then I looked at his publications and … there was one that caught my attention much more than the others, because I was in it and I didn’t know what that publication was. I was the second author of a publication that I had never read (participant 14). I saw a colleague who had plagiarized another colleague. [When the colleague] found out about it, he complained. So, plagiarism is a serious [ethical breach]. I would also say that there is a certain amount of competition in the university faculties, especially for grants (…). There are people who want to win at all costs or get as much as possible. They are not necessarily going to consider their colleagues. They don’t have much of a collegial spirit (participant 10).
These examples of research misbehaviour or misconduct are sometimes due to or associated with situations of conflicts of interest, which may be poorly managed by certain researchers or research teams, as noted by many participants.
Conflict of interest
The actors and institutions involved in research have diverse interests, like all humans and institutions. As noted in Chap. 7 of the Canadian Tri-Council Policy Statement: Ethical Conduct for Research Involving Humans (TCPS2, 2018),
“researchers and research students hold trust relationships, either directly or indirectly, with participants, research sponsors, institutions, their professional bodies and society. These trust relationships can be put at risk by conflicts of interest that may compromise independence, objectivity or ethical duties of loyalty. Although the potential for such conflicts has always existed, pressures on researchers (i.e., to delay or withhold dissemination of research outcomes or to use inappropriate recruitment strategies) heighten concerns that conflicts of interest may affect ethical behaviour” (p. 92).
The sources of these conflicts are varied and can include interpersonal conflicts, financial partnerships, third-party pressures, academic or economic interests, a researcher holding multiple roles within an institution, or any other incentive that may compromise a researcher’s independence, integrity, and neutrality (TCPS2, 2018). While it is not possible to eliminate all conflicts of interest, it is important to manage them properly and to avoid temptations to behave unethically.
Ethical temptations correspond to situations in which people are tempted to prioritize their own interests to the detriment of the ethical goods that should, in their own context, govern their actions (Swisher et al., 2005 ). In the case of researchers, this refers to situations that undermine independence, integrity, neutrality, or even the set of principles that govern research ethics (TCPS2, 2018) or the responsible conduct of research. According to study participants, these types of ethical issues frequently occur in research. Many participants, especially researchers and REB members, reported that conflicts of interest can arise when members of an organization make decisions to obtain large financial rewards or to increase their academic profile, often at the expense of the interests of members of their research team, research participants, or even the populations affected by their research.
A company that puts money into making its drug work wants its drug to work. So, homeopathy is a good example, because there are not really any consequences of homeopathy, there are not very many side effects, because there are no effects at all. So, it’s not dangerous, but it’s not a good treatment either. But some people will want to make it work. And that’s a big issue when you’re sitting at a table and there are eight researchers, and there are two or three who are like that, and then there are four others who are neutral, and I say to myself, this is not science. I think that this is a very big ethical issue (participant 14). There are also times in some research where there will be more links with pharmaceutical companies. Obviously, there are then large amounts of money that will be very interesting for the health-care institutions because they still receive money for clinical trials. They’re still getting some compensation because its time consuming for the people involved and all that. The pharmaceutical companies have money, so they will compensate, and that is sometimes interesting for the institutions, and since we are a bit caught up in this, in the sense that we have no choice but to accept it. (…) It may not be the best research in the world, there may be a lot of side effects due to the drugs, but it’s good to accept it, we’re going to be part of the clinical trial (participant 3). It is integrity, what we believe should be done or said. Often by the pressure of the environment, integrity is in tension with the pressures of the environment, so it takes resistance, it takes courage in research. (…) There were all the debates there about the problems of research that was funded and then the companies kept control over what was written. That was really troubling for a lot of researchers (participant 5).
Further, these situations sometimes have negative consequences for research participants as reported by some participants.
Respect for research participants
Many research projects, whether they are psychosocial or biomedical in nature, involve human participants. Relationships between the members of research teams and their research participants raise ethical issues that can be complex. Research projects must always be designed to respect the rights and interests of research participants, and not just those of researchers. However, participants in our study – i.e., REB members, researchers, and research ethics experts – noted that some research teams seem to put their own interests ahead of those of research participants. They also emphasized the importance of ensuring the respect, well-being, and safety of research participants. The ethical issues related to this unit of meaning are: respect for free, informed and ongoing consent of research participants; respect for and the well-being of participants; data protection and confidentiality; over-solicitation of participants; ownership of the data collected on participants; the sometimes high cost of scientific innovations and their accessibility; balance between the social benefits of research and the risks to participants (particularly in terms of safety); balance between collective well-being (development of knowledge) and the individual rights of participants; exploitation of participants; paternalism when working with populations in vulnerable situations; and the social acceptability of certain types of research. The following excerpts present some of these issues.
Where it disturbs me ethically is in the medical field – because it’s more in the medical field that we’re going to see this – when consent forms are presented to patients to solicit them as participants, and then [these forms] have an average of 40 pages. That annoys me. When they say that it has to be easy to understand and all that, adapted to the language, and then the hyper-technical language plus there are 40 pages to read, I don’t understand how you’re going to get informed consent after reading 40 pages. (…) For me, it doesn’t work. I read them to evaluate them and I have a certain level of education and experience in ethics, and there are times when I don’t understand anything (participant 2). There is a lot of pressure from researchers who want to recruit research participants (…). The idea that when you enter a health care institution, you become a potential research participant, when you say “yes to a research, you check yes to all research”, then everyone can ask you. I think that researchers really have this fantasy of saying to themselves: “as soon as people walk through the door of our institution, they become potential participants with whom we can communicate and get them involved in all projects”. There’s a kind of idea that, yes, it can be done, but it has to be somewhat supervised to avoid over-solicitation (…). Researchers are very interested in facilitating recruitment and making it more fluid, but perhaps to the detriment of confidentiality, privacy, and respect; sometimes that’s what it is, to think about what type of data you’re going to have in your bank of potential participants? Is it just name and phone number or are you getting into more sensitive information? (participant 9).
In addition, one participant reported that their university does not provide the resources required to respect the confidentiality of research participants.
The issue is as follows: researchers, of course, commit to protecting data with passwords and all that, but we realize that in practice, it is more difficult. It is not always as protected as one might think, because professor-researchers will run out of space. Will the universities make rooms available to researchers, places where they can store these things, especially when they have paper documentation, and is there indeed a guarantee of confidentiality? Some researchers have told me: “Listen; there are even filing cabinets in the corridors”. So, that certainly poses a concrete challenge. How do we go about challenging the administrative authorities? Tell them it’s all very well to have an ethics committee, but you have to help us, you also have to make sure that the necessary infrastructures are in place so that what we are proposing is really put into practice (participant 4).
If the relationships with research participants are likely to raise ethical issues, so too are the relationships with students, notably research assistants. On this topic, several participants discussed the lack of supervision or recognition offered to research assistants by researchers as well as the power imbalances between members of the research team.
Lack of Supervision and Power Imbalances
Many research teams are composed not only of researchers, but also of students who work as research assistants. The relationship between research assistants and other members of research teams can sometimes be problematic and raise ethical issues, particularly because of the inevitable power asymmetries. In the context of this study, several participants – including a research assistant, REB members, and researchers – discussed the lack of supervision or recognition of the work carried out by students, psychological pressure, and the more or less well-founded promises that are sometimes made to students. Participants also mentioned the exploitation of students by certain research teams, which manifest when students are inadequately paid, i.e., not reflective of the number of hours actually worked, not a fair wage, or even a wage at all.
[As a research assistant], it was more of a feeling of distress that I felt then because I didn’t know what to do. (…) I was supposed to get coaching or be supported, but I didn’t get anything in the end. It was like, “fix it by yourself”. (…) All research assistants were supposed to be supervised, but in practice they were not (participant 1). Very often, we have a master’s or doctoral student that we put on a subject and we consider that the project will be well done, while the student is learning. So, it happens that the student will do a lot of work and then we realize that the work is poorly done, and it is not necessarily the student’s fault. He wasn’t necessarily well supervised. There are directors who have 25 students, and they just don’t supervise them (participant 14). I think it’s really the power relationship. I thought to myself, how I saw my doctorate, the beginning of my research career, I really wanted to be in that laboratory, but they are the ones who are going to accept me or not, so what do I do to be accepted? I finally accept their conditions [which was to work for free]. If these are the conditions that are required to enter this lab, I want to go there. So, what do I do, well I accepted. It doesn’t make sense, but I tell myself that I’m still privileged, because I don’t have so many financial worries, one more reason to work for free, even though it doesn’t make sense (participant 1). In research, we have research assistants. (…). The fact of using people… so that’s it, you have to take into account where they are, respect them, but at the same time they have to show that they are there for the research. In English, we say “carry” or take care of people. With research assistants, this is often a problem that I have observed: for grant machines, the person is the last to be found there. Researchers, who will take, use student data, without giving them the recognition for it (participant 5). The problem at our university is that they reserve funding for Canadian students. The doctoral clientele in my field is mostly foreign students. So, our students are poorly funded. I saw one student end up in the shelter, in a situation of poverty. It ended very badly for him because he lacked financial resources. Once you get into that dynamic, it’s very hard to get out. I was made aware of it because the director at the time had taken him under her wing and wanted to try to find a way to get him out of it. So, most of my students didn’t get funded (participant 16). There I wrote “manipulation”, but it’s kind of all promises all the time. I, for example, was promised a lot of advancement, like when I got into the lab as a graduate student, it was said that I had an interest in [this particular area of research]. I think there are a lot of graduate students who must have gone through that, but it is like, “Well, your CV has to be really good, if you want to do a lot of things and big things. If you do this, if you do this research contract, the next year you could be the coordinator of this part of the lab and supervise this person, get more contracts, be paid more. Let’s say: you’ll be invited to go to this conference, this big event”. They were always dangling something, but you have to do that first to get there. But now, when you’ve done that, you have to do this business. It’s like a bit of manipulation, I think. That was very hard to know who is telling the truth and who is not (participant 1).
These ethical issues have significant negative consequences for students. Indeed, they sometimes find themselves at the mercy of researchers, for whom they work, struggling to be recognized and included as authors of an article, for example, or to receive the salary that they are due. For their part, researchers also sometimes find themselves trapped in research structures that can negatively affect their well-being. As many participants reported, researchers work in organizations that set very high productivity standards and in highly competitive contexts, all within a general culture characterized by individualism.
Individualism and performance
Participants, especially researchers, discussed the culture of individualism and performance that characterizes the academic environment. In glorifying excellence, some universities value performance and productivity, often at the expense of psychological well-being and work-life balance (i.e., work overload and burnout). Participants noted that there are ethical silences in their organizations on this issue, and that the culture of individualism and performance is not challenged for fear of retribution or simply to survive, i.e., to perform as expected. Participants felt that this culture can have a significant negative impact on the quality of the research conducted, as research teams try to maximize the quantity of their work (instead of quality) in a highly competitive context, which is then exacerbated by a lack of resources and support, and where everything must be done too quickly.
The work-life balance with the professional ethics related to work in a context where you have too much and you have to do a lot, it is difficult to balance all that and there is a lot of pressure to perform. If you don’t produce enough, that’s it; after that, you can’t get any more funds, so that puts pressure on you to do more and more and more (participant 3). There is a culture, I don’t know where it comes from, and that is extremely bureaucratic. If you dare to raise something, you’re going to have many, many problems. They’re going to make you understand it. So, I don’t talk. It is better: your life will be easier. I think there are times when you have to talk (…) because there are going to be irreparable consequences. (…) I’m not talking about a climate of terror, because that’s exaggerated, it’s not true, people are not afraid. But people close their office door and say nothing because it’s going to make their work impossible and they’re not going to lose their job, they’re not going to lose money, but researchers need time to be focused, so they close their office door and say nothing (participant 16).
Researchers must produce more and more, and they feel little support in terms of how to do such production, ethically, and how much exactly they are expected to produce. As this participant reports, the expectation is an unspoken rule: more is always better.
It’s sometimes the lack of a clear line on what the expectations are as a researcher, like, “ah, we don’t have any specific expectations, but produce, produce, produce, produce.” So, in that context, it’s hard to be able to put the line precisely: “have I done enough for my work?” (participant 3).
Inadequate ethical Guidance
While the productivity expectation is not clear, some participants – including researchers, research ethics experts, and REB members – also felt that the ethical expectations of some REBs were unclear. The issue of the inadequate ethical guidance of research includes the administrative mechanisms to ensure that research projects respect the principles of research ethics. According to those participants, the forms required for both researchers and REB members are increasingly long and numerous, and one participant noted that the standards to be met are sometimes outdated and disconnected from the reality of the field. Multicentre ethics review (by several REBs) was also critiqued by a participant as an inefficient method that encumbers the processes for reviewing research projects. Bureaucratization imposes an ever-increasing number of forms and ethics guidelines that actually hinder researchers’ ethical reflection on the issues at stake, leading the ethics review process to be perceived as purely bureaucratic in nature.
The ethical dimension and the ethical review of projects have become increasingly bureaucratized. (…) When I first started working (…) it was less bureaucratic, less strict then. I would say [there are now] tons of forms to fill out. Of course, we can’t do without it, it’s one of the ways of marking out ethics and ensuring that there are ethical considerations in research, but I wonder if it hasn’t become too bureaucratized, so that it’s become a kind of technical reflex to fill out these forms, and I don’t know if people really do ethical reflection as such anymore (participant 10). The fundamental structural issue, I would say, is the mismatch between the normative requirements and the real risks posed by the research, i.e., we have many, many requirements to meet; we have very long forms to fill out but the research projects we evaluate often pose few risks (participant 8). People [in vulnerable situations] were previously unable to participate because of overly strict research ethics rules that were to protect them, but in the end [these rules] did not protect them. There was a perverse effect, because in the end there was very little research done with these people and that’s why we have very few results, very little evidence [to support practices with these populations] so it didn’t improve the quality of services. (…) We all understand that we have to be careful with that, but when the research is not too risky, we say to ourselves that it would be good because for once a researcher who is interested in that population, because it is not a very popular population, it would be interesting to have results, but often we are blocked by the norms, and then we can’t accept [the project] (participant 2).
Moreover, as one participant noted, accessing ethics training can be a challenge.
There is no course on research ethics. […] Then, I find that it’s boring because you go through university and you come to do your research and you know how to do quantitative and qualitative research, but all the research ethics, where do you get this? I don’t really know (participant 13).
Yet, such training could provide relevant tools to resolve, to some extent, the ethical issues that commonly arise in research. That said, and as noted by many participants, many ethical issues in research are related to social injustices over which research actors have little influence.
Social Injustices
For many participants, notably researchers, the issues that concern social injustices are those related to power asymmetries, stigma, or issues of equity, diversity, and inclusion, i.e., social injustices related to people’s identities (Blais & Drolet, 2022 ). Participants reported experiencing or witnessing discrimination from peers, administration, or lab managers. Such oppression is sometimes cross-sectional and related to a person’s age, cultural background, gender or social status.
I have my African colleague who was quite successful when he arrived but had a backlash from colleagues in the department. I think it’s unconscious, nobody is overtly racist. But I have a young person right now who is the same, who has the same success, who got exactly the same early career award and I don’t see the same backlash. He’s just as happy with what he’s doing. It’s normal, they’re young and they have a lot of success starting out. So, I think there is discrimination. Is it because he is African? Is it because he is black? I think it’s on a subconscious level (participant 16).
Social injustices were experienced or reported by many participants, and included issues related to difficulties in obtaining grants or disseminating research results in one’s native language (i.e., even when there is official bilingualism) or being considered credible and fundable in research when one researcher is a woman.
If you do international research, there are things you can’t talk about (…). It is really a barrier to research to not be able to (…) address this question [i.e. the question of inequalities between men and women]. Women’s inequality is going to be addressed [but not within the country where the research takes place as if this inequality exists elsewhere but not here]. There are a lot of women working on inequality issues, doing work and it’s funny because I was talking to a young woman who works at Cairo University and she said to me: “Listen, I saw what you had written, you’re right. I’m willing to work on this but guarantee me a position at your university with a ticket to go”. So yes, there are still many barriers [for women in research] (participant 16).
Because of the varied contextual characteristics that intervene in their occurrence, these social injustices are also related to distributive injustices, as discussed by many participants.
Distributive Injustices
Although there are several views of distributive justice, a classical definition such as that of Aristotle ( 2012 ), describes distributive justice as consisting in distributing honours, wealth, and other social resources or benefits among the members of a community in proportion to their alleged merit. Justice, then, is about determining an equitable distribution of common goods. Contemporary theories of distributive justice are numerous and varied. Indeed, many authors (e.g., Fraser 2011 ; Mills, 2017 ; Sen, 2011 ; Young, 2011 ) have, since Rawls ( 1971 ), proposed different visions of how social burdens and benefits should be shared within a community to ensure equal respect, fairness, and distribution. In our study, what emerges from participants’ narratives is a definite concern for this type of justice. Women researchers, francophone researchers, early career researchers or researchers belonging to racialized groups all discussed inequities in the distribution of research grants and awards, and the extra work they need to do to somehow prove their worth. These inequities are related to how granting agencies determine which projects will be funded.
These situations make me work 2–3 times harder to prove myself and to show people in power that I have a place as a woman in research (participant 12). Number one: it’s conservative thinking. The older ones control what comes in. So, the younger people have to adapt or they don’t get funded (participant 14).
Whether it is discrimination against stigmatized or marginalized populations or interest in certain hot topics, granting agencies judge research projects according to criteria that are sometimes questionable, according to those participants. Faced with difficulties in obtaining funding for their projects, several strategies – some of which are unethical – are used by researchers in order to cope with these situations.
Sometimes there are subjects that everyone goes to, such as nanotechnology (…), artificial intelligence or (…) the therapeutic use of cannabis, which are very fashionable, and this is sometimes to the detriment of other research that is just as relevant, but which is (…), less sexy, less in the spirit of the time. (…) Sometimes this can lead to inequities in the funding of certain research sectors (participant 9). When we use our funds, we get them given to us, we pretty much say what we think we’re going to do with them, but things change… So, when these things change, sometimes it’s an ethical decision, but by force of circumstances I’m obliged to change the project a little bit (…). Is it ethical to make these changes or should I just let the money go because I couldn’t use it the way I said I would? (participant 3).
Moreover, these distributional injustices are not only linked to social injustices, but also epistemic injustices. Indeed, the way in which research honours and grants are distributed within the academic community depends on the epistemic authority of the researchers, which seems to vary notably according to their language of use, their age or their gender, but also to the research design used (inductive versus deductive), their decision to use (or not use) animals in research, or to conduct activist research.
Epistemic injustices
The philosopher Fricker ( 2007 ) conceptualized the notions of epistemic justice and injustice. Epistemic injustice refers to a form of social inequality that manifests itself in the access, recognition, and production of knowledge as well as the various forms of ignorance that arise (Godrie & Dos Santos, 2017 ). Addressing epistemic injustice necessitates acknowledging the iniquitous wrongs suffered by certain groups of socially stigmatized individuals who have been excluded from knowledge, thus limiting their abilities to interpret, understand, or be heard and account for their experiences. In this study, epistemic injustices were experienced or reported by some participants, notably those related to difficulties in obtaining grants or disseminating research results in one’s native language (i.e., even when there is official bilingualism) or being considered credible and fundable in research when a researcher is a woman or an early career researcher.
I have never sent a grant application to the federal government in English. I have always done it in French, even though I know that when you receive the review, you can see that reviewers didn’t understand anything because they are English-speaking. I didn’t want to get in the boat. It’s not my job to translate, because let’s be honest, I’m not as good in English as I am in French. So, I do them in my first language, which is the language I’m most used to. Then, technically at the administrative level, they are supposed to be able to do it, but they are not good in French. (…) Then, it’s a very big Canadian ethical issue, because basically there are technically two official languages, but Canada is not a bilingual country, it’s a country with two languages, either one or the other. (…) So I was not funded (participant 14).
Researchers who use inductive (or qualitative) methods observed that their projects are sometimes less well reviewed or understood, while research that adopts a hypothetical-deductive (or quantitative) or mixed methods design is better perceived, considered more credible and therefore more easily funded. Of course, regardless of whether a research project adopts an inductive, deductive or mixed-methods scientific design, or whether it deals with qualitative or quantitative data, it must respect a set of scientific criteria. A research project should achieve its objectives by using proven methods that, in the case of inductive research, are credible, reliable, and transferable or, in the case of deductive research, generalizable, objective, representative, and valid (Drolet & Ruest, accepted ). Participants discussing these issues noted that researchers who adopt a qualitative design or those who question the relevance of animal experimentation or are not militant have sometimes been unfairly devalued in their epistemic authority.
There is a mini war between quantitative versus qualitative methods, which I think is silly because science is a method. If you apply the method well, it doesn’t matter what the field is, it’s done well and it’s perfect ” (participant 14). There is also the issue of the place of animals in our lives, because for me, ethics is human ethics, but also animal ethics. Then, there is a great evolution in society on the role of the animal… with the new law that came out in Quebec on the fact that animals are sensitive beings. Then, with the rise of the vegan movement, [we must ask ourselves]: “Do animals still have a place in research?” That’s a big question and it also means that there are practices that need to evolve, but sometimes there’s a disconnection between what’s expected by research ethics boards versus what’s expected in the field (participant 15). In research today, we have more and more research that is militant from an ideological point of view. And so, we have researchers, because they defend values that seem important to them, we’ll talk for example about the fight for equality and social justice. They have pressure to defend a form of moral truth and have the impression that everyone thinks like them or should do so, because they are defending a moral truth. This is something that we see more and more, namely the lack of distance between ideology and science (participant 8).
The combination or intersectionality of these inequities, which seems to be characterized by a lack of ethical support and guidance, is experienced in the highly competitive and individualistic context of research; it provides therefore the perfect recipe for researchers to experience ethical distress.
Ethical distress
The concept of “ethical distress” refers to situations in which people know what they should do to act ethically, but encounter barriers, generally of an organizational or systemic nature, limiting their power to act according to their moral or ethical values (Drolet & Ruest, 2021 ; Jameton, 1984 ; Swisher et al., 2005 ). People then run the risk of finding themselves in a situation where they do not act as their ethical conscience dictates, which in the long term has the potential for exhaustion and distress. The examples reported by participants in this study point to the fact that researchers in particular may be experiencing significant ethical distress. This distress takes place in a context of extreme competition, constant injunctions to perform, and where administrative demands are increasingly numerous and complex to complete, while paradoxically, they lack the time to accomplish all their tasks and responsibilities. Added to these demands are a lack of resources (human, ethical, and financial), a lack of support and recognition, and interpersonal conflicts.
We are in an environment, an elite one, you are part of it, you know what it is: “publish or perish” is the motto. Grants, there is a high level of performance required, to do a lot, to publish, to supervise students, to supervise them well, so yes, it is clear that we are in an environment that is conducive to distress. (…). Overwork, definitely, can lead to distress and eventually to exhaustion. When you know that you should take the time to read the projects before sharing them, but you don’t have the time to do that because you have eight that came in the same day, and then you have others waiting… Then someone rings a bell and says: “ah but there, the protocol is a bit incomplete”. Oh yes, look at that, you’re right. You make up for it, but at the same time it’s a bit because we’re in a hurry, we don’t necessarily have the resources or are able to take the time to do things well from the start, we have to make up for it later. So yes, it can cause distress (participant 9). My organization wanted me to apply in English, and I said no, and everyone in the administration wanted me to apply in English, and I always said no. Some people said: “Listen, I give you the choice”, then some people said: “Listen, I agree with you, but if you’re not [submitting] in English, you won’t be funded”. Then the fact that I am young too, because very often they will look at the CV, they will not look at the project: “ah, his CV is not impressive, we will not finance him”. This is complete nonsense. The person is capable of doing the project, the project is fabulous: we fund the project. So, that happened, organizational barriers: that happened a lot. I was not eligible for Quebec research funds (…). I had big organizational barriers unfortunately (participant 14). At the time of my promotion, some colleagues were not happy with the type of research I was conducting. I learned – you learn this over time when you become friends with people after you enter the university – that someone was against me. He had another candidate in mind, and he was angry about the selection. I was under pressure for the first three years until my contract was renewed. I almost quit at one point, but another colleague told me, “No, stay, nothing will happen”. Nothing happened, but these issues kept me awake at night (participant 16).
This difficult context for many researchers affects not only the conduct of their own research, but also their participation in research. We faced this problem in our study, despite the use of multiple recruitment methods, including more than 200 emails – of which 191 were individual solicitations – sent to potential participants by the two research assistants. REB members and organizations overseeing or supporting research (n = 17) were also approached to see if some of their employees would consider participating. While it was relatively easy to recruit REB members and research ethics experts, our team received a high number of non-responses to emails (n = 175) and some refusals (n = 5), especially by researchers. The reasons given by those who replied were threefold: (a) fear of being easily identified should they take part in the research, (b) being overloaded and lacking time, and (c) the intrusive aspect of certain questions (i.e., “Have you experienced a burnout episode? If so, have you been followed up medically or psychologically?”). In light of these difficulties and concerns, some questions in the socio-demographic questionnaire were removed or modified. Talking about burnout in research remains a taboo for many researchers, which paradoxically can only contribute to the unresolved problem of unhealthy research environments.
Returning to the research question and objective
The question that prompted this research was: What are the ethical issues in research? The purpose of the study was to describe these issues from the perspective of researchers (from different disciplines), research ethics board (REB) members, and research ethics experts. The previous section provided a detailed portrait of the ethical issues experienced by different research stakeholders: these issues are numerous, diverse and were recounted by a range of stakeholders.
The results of the study are generally consistent with the literature. For example, as in our study, the literature discusses the lack of research integrity on the part of some researchers (Al-Hidabi et al., 2018 ; Swazey et al., 1993 ), the numerous conflicts of interest experienced in research (Williams-Jones et al., 2013 ), the issues of recruiting and obtaining the free and informed consent of research participants (Provencher et al., 2014 ; Keogh & Daly, 2009 ), the sometimes difficult relations between researchers and REBs (Drolet & Girard, 2020 ), the epistemological issues experienced in research (Drolet & Ruest, accepted; Sieber 2004 ), as well as the harmful academic context in which researchers evolve, insofar as this is linked to a culture of performance, an overload of work in a context of accountability (Berg & Seeber, 2016 ; FQPPU; 2019 ) that is conducive to ethical distress and even burnout.
If the results of the study are generally in line with those of previous publications on the subject, our findings also bring new elements to the discussion while complementing those already documented. In particular, our results highlight the role of systemic injustices – be they social, distributive or epistemic – within the environments in which research is carried out, at least in Canada. To summarize, the results of our study point to the fact that the relationships between researchers and research participants are likely still to raise worrying ethical issues, despite widely accepted research ethics norms and institutionalized review processes. Further, the context in which research is carried out is not only conducive to breaches of ethical norms and instances of misbehaviour or misconduct, but also likely to be significantly detrimental to the health and well-being of researchers, as well as research assistants. Another element that our research also highlighted is the instrumentalization and even exploitation of students and research assistants, which is another important and worrying social injustice given the inevitable power imbalances between students and researchers.
Moreover, in a context in which ethical issues are often discussed from a micro perspective, our study helps shed light on both the micro- and macro-level ethical dimensions of research (Bronfenbrenner, 1979 ; Glaser 1994 ). However, given that ethical issues in research are not only diverse, but also and above all complex, a broader perspective that encompasses the interplay between the micro and macro dimensions can enable a better understanding of these issues and thereby support the identification of the multiple factors that may be at their origin. Triangulating the perspectives of researchers with those of REB members and research ethics experts enabled us to bring these elements to light, and thus to step back from and critique the way that research is currently conducted. To this end, attention to socio-political elements such as the performance culture in academia or how research funds are distributed, and according to what explicit and implicit criteria, can contribute to identifying the sources of the ethical issues described above.
Contemporary culture characterized by the social acceleration
The German sociologist and philosopher Rosa (2010) argues that late modernity – that is, the period between the 1980s and today – is characterized by a phenomenon of social acceleration that causes various forms of alienation in our relationship to time, space, actions, things, others and ourselves. Rosa distinguishes three types of acceleration: technical acceleration , the acceleration of social changes and the acceleration of the rhythm of life . According to Rosa, social acceleration is the main problem of late modernity, in that the invisible social norm of doing more and faster to supposedly save time operates unchallenged at all levels of individual and collective life, as well as organizational and social life. Although we all, researchers and non-researchers alike, perceive this unspoken pressure to be ever more productive, the process of social acceleration as a new invisible social norm is our blind spot, a kind of tyrant over which we have little control. This conceptualization of the contemporary culture can help us to understand the context in which research is conducted (like other professional practices). To this end, Berg & Seeber ( 2016 ) invite faculty researchers to slow down in order to better reflect and, in the process, take care of their health and their relationships with their colleagues and students. Many women professors encourage their fellow researchers, especially young women researchers, to learn to “say No” in order to protect their mental and physical health and to remain in their academic careers (Allaire & Descheneux, 2022 ). These authors also remind us of the relevance of Kahneman’s ( 2012 ) work which demonstrates that it takes time to think analytically, thoroughly, and logically. Conversely, thinking quickly exposes humans to cognitive and implicit biases that then lead to errors in thinking (e.g., in the analysis of one’s own research data or in the evaluation of grant applications or student curriculum vitae). The phenomenon of social acceleration, which pushes the researcher to think faster and faster, is likely to lead to unethical bad science that can potentially harm humankind. In sum, Rosa’s invitation to contemporary critical theorists to seriously consider the problem of social acceleration is particularly insightful to better understand the ethical issues of research. It provides a lens through which to view the toxic context in which research is conducted today, and one that was shared by the participants in our study.
Clark & Sousa ( 2022 ) note, it is important that other criteria than the volume of researchers’ contributions be valued in research, notably quality. Ultimately, it is the value of the knowledge produced and its influence on the concrete lives of humans and other living beings that matters, not the quantity of publications. An interesting articulation of this view in research governance is seen in a change in practice by Australia’s national health research funder: they now restrict researchers to listing on their curriculum vitae only the top ten publications from the past ten years (rather than all of their publications), in order to evaluate the quality of contributions rather than their quantity. To create environments conducive to the development of quality research, it is important to challenge the phenomenon of social acceleration, which insidiously imposes a quantitative normativity that is both alienating and detrimental to the quality and ethical conduct of research. Based on our experience, we observe that the social norm of acceleration actively disfavours the conduct of empirical research on ethics in research. The fact is that researchers are so busy that it is almost impossible for them to find time to participate in such studies. Further, operating in highly competitive environments, while trying to respect the values and ethical principles of research, creates ethical paradoxes for members of the research community. According to Malherbe ( 1999 ), an ethical paradox is a situation where an individual is confronted by contradictory injunctions (i.e., do more, faster, and better). And eventually, ethical paradoxes lead individuals to situations of distress and burnout, or even to ethical failures (i.e., misbehaviour or misconduct) in the face of the impossibility of responding to contradictory injunctions.
Strengths and Limitations of the study
The triangulation of perceptions and experiences of different actors involved in research is a strength of our study. While there are many studies on the experiences of researchers, rarely are members of REBs and experts in research ethics given the space to discuss their views of what are ethical issues. Giving each of these stakeholders a voice and comparing their different points of view helped shed a different and complementary light on the ethical issues that occur in research. That said, it would have been helpful to also give more space to issues experienced by students or research assistants, as the relationships between researchers and research assistants are at times very worrying, as noted by a participant, and much work still needs to be done to eliminate the exploitative situations that seem to prevail in certain research settings. In addition, no Indigenous or gender diverse researchers participated in the study. Given the ethical issues and systemic injustices that many people from these groups face in Canada (Drolet & Goulet, 2018 ; Nicole & Drolet, in press ), research that gives voice to these researchers would be relevant and contribute to knowledge development, and hopefully also to change in research culture.
Further, although most of the ethical issues discussed in this article may be transferable to the realities experienced by researchers in other countries, the epistemic injustice reported by Francophone researchers who persist in doing research in French in Canada – which is an officially bilingual country but in practice is predominantly English – is likely specific to the Canadian reality. In addition, and as mentioned above, recruitment proved exceedingly difficult, particularly amongst researchers. Despite this difficulty, we obtained data saturation for all but two themes – i.e., exploitation of students and ethical issues of research that uses animals. It follows that further empirical research is needed to improve our understanding of these specific issues, as they may diverge to some extent from those documented here and will likely vary across countries and academic research contexts.
Conclusions
This study, which gave voice to researchers, REB members, and ethics experts, reveals that the ethical issues in research are related to several problematic elements as power imbalances and authority relations. Researchers and research assistants are subject to external pressures that give rise to integrity issues, among others ethical issues. Moreover, the current context of social acceleration influences the definition of the performance indicators valued in academic institutions and has led their members to face several ethical issues, including social, distributive, and epistemic injustices, at different steps of the research process. In this study, ten categories of ethical issues were identified, described and illustrated: (1) research integrity, (2) conflicts of interest, (3) respect for research participants, (4) lack of supervision and power imbalances, (5) individualism and performance, (6) inadequate ethical guidance, (7) social injustices, (8) distributive injustices, (9) epistemic injustices, and (10) ethical distress. The triangulation of the perspectives of different members (i.e., researchers from different disciplines, REB members, research ethics experts, and one research assistant) involved in the research process made it possible to lift the veil on some of these ethical issues. Further, it enabled the identification of additional ethical issues, especially systemic injustices experienced in research. To our knowledge, this is the first time that these injustices (social, distributive, and epistemic injustices) have been clearly identified.
Finally, this study brought to the fore several problematic elements that are important to address if the research community is to develop and implement the solutions needed to resolve the diverse and transversal ethical issues that arise in research institutions. A good starting point is the rejection of the corollary norms of “publish or perish” and “do more, faster, and better” and their replacement with “publish quality instead of quantity”, which necessarily entails “do less, slower, and better”. It is also important to pay more attention to the systemic injustices within which researchers work, because these have the potential to significantly harm the academic careers of many researchers, including women researchers, early career researchers, and those belonging to racialized groups as well as the health, well-being, and respect of students and research participants.
Acknowledgements
The team warmly thanks the participants who took part in the research and who made this study possible. Marie-Josée Drolet thanks the five research assistants who participated in the data collection and analysis: Julie-Claude Leblanc, Élie Beauchemin, Pénéloppe Bernier, Louis-Pierre Côté, and Eugénie Rose-Derouin, all students at the Université du Québec à Trois-Rivières (UQTR), two of whom were active in the writing of this article. MJ Drolet and Bryn Williams-Jones also acknowledge the financial contribution of the Social Sciences and Humanities Research Council of Canada (SSHRC), which supported this research through a grant. We would also like to thank the reviewers of this article who helped us improve it, especially by clarifying and refining our ideas.
Competing Interests and Funding
As noted in the Acknowledgements, this research was supported financially by the Social Sciences and Humanities Research Council of Canada (SSHRC).
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
- Al-Hidabi, Abdulmalek, M. D., & The, P. L. (2018). Multiple Publications: The Main Reason for the Retraction of Papers in Computer Science. In K. Arai, S. Kapoor, & R. Bhatia (eds), Future of Information and Communication Conference (FICC): Advances in Information and Communication, Advances in Intelligent Systems and Computing (AISC), Springer, vol. 886, pp. 511–526
- Allaire, S., & Deschenaux, F. (2022). Récits de professeurs d’université à mi-carrière. Si c’était à refaire… . Presses de l’Université du Québec
- Aristotle . Aristotle’s Nicomachean Ethics. Chicago: The University of Chicago Press; 2012. [ Google Scholar ]
- Bahn S. Keeping Academic Field Researchers Safe: Ethical Safeguards. Journal of Academic Ethics. 2012; 10 :83–91. doi: 10.1007/s10805-012-9159-2. [ CrossRef ] [ Google Scholar ]
- Balk DE. Bereavement Research Using Control Groups: Ethical Obligations and Questions. Death Studies. 1995; 19 :123–138. doi: 10.1080/07481189508252720. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Beauchemin, É., Côté, L. P., Drolet, M. J., & Williams-Jones, B. (2021). Conceptualizing Ethical Issues in the Conduct of Research: Results from a Critical and Systematic Literature Review. Journal of Academic Ethics , Early Online. 10.1007/s10805-021-09411-7
- Berg, M., & Seeber, B. K. (2016). The Slow Professor . University of Toronto Press
- Birchley G, Huxtable R, Murtagh M, Meulen RT, Flach P, Gooberman-Hill R. Smart homes, private homes? An empirical study of technology researchers’ perceptions of ethical issues in developing smart-home health technologies. BMC Medical Ethics. 2017; 18 (23):1–13. doi: 10.1186/s12910-017-0183-z. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Blais, J., & Drolet, M. J. (2022). Les injustices sociales vécues en camp de réfugiés: les comprendre pour mieux intervenir auprès de personnes ayant séjourné dans un camp de réfugiés. Recueil annuel belge d’ergothérapie , 14, 37–48
- Bogdan, R. C., & Biklen, S. K. (2006). Qualitative research in education: An introduction to theory and methods . Allyn & Bacon
- Bouffard C. Le développement des pratiques de la génétique médicale et la construction des normes bioéthiques. Anthropologie et Sociétés. 2000; 24 (2):73–90. doi: 10.7202/015650ar. [ CrossRef ] [ Google Scholar ]
- Bronfenbrenner, U. (1979). The Ecology of Human development. Experiments by nature and design . Harvard University Press
- Bruhn JG, Zajac G, Al-Kazemi AA, Prescott LD. Moral positions and academic conduct: Parameters of tolerance for ethics failure. Journal of Higher Education. 2002; 73 (4):461–493. doi: 10.1353/jhe.2002.0033. [ CrossRef ] [ Google Scholar ]
- Clark, A., & Sousa (2022). It’s time to end Canada’s obsession with research quantity. University Affairs/Affaires universitaires , February 14th. https://www.universityaffairs.ca/career-advice/effective-successfull-happy-academic/its-time-to-end-canadas-obsession-with-research-quantity/?utm_source=University+Affairs+e-newsletter&utm_campaign=276a847f 70-EMAIL_CAMPAIGN_2022_02_16&utm_medium=email&utm_term=0_314bc2ee29-276a847f70-425259989
- Colnerud G. Ethical dilemmas in research in relation to ethical review: An empirical study. Research Ethics. 2015; 10 (4):238–253. doi: 10.1177/1747016114552339. [ CrossRef ] [ Google Scholar ]
- Davison J. Dilemmas in Research: Issues of Vulnerability and Disempowerment for the Social Workers/Researcher. Journal of Social Work Practice. 2004; 18 (3):379–393. doi: 10.1080/0265053042000314447. [ CrossRef ] [ Google Scholar ]
- DePoy E, Gitlin LN. Introduction to Research. St. Louis: Elsevier Mosby; 2010. [ Google Scholar ]
- Drolet, M. J., & Goulet, M. (2018). Travailler avec des patients autochtones du Canada ? Perceptions d’ergothérapeutes du Québec des enjeux éthiques de cette pratique. Recueil annuel belge francophone d’ergothérapie , 10 , 25–56
- Drolet MJ, Girard K. Les enjeux éthiques de la recherche en ergothérapie: un portrait préoccupant. Revue canadienne de bioéthique. 2020; 3 (3):21–40. doi: 10.7202/1073779ar. [ CrossRef ] [ Google Scholar ]
- Drolet MJ, Girard K, Gaudet R. Les enjeux éthiques de l’enseignement en ergothérapie: des injustices au sein des départements universitaires. Revue canadienne de bioéthique. 2020; 3 (1):22–36. [ Google Scholar ]
- Drolet MJ, Maclure J. Les enjeux éthiques de la pratique de l’ergothérapie: perceptions d’ergothérapeutes. Revue Approches inductives. 2016; 3 (2):166–196. doi: 10.7202/1037918ar. [ CrossRef ] [ Google Scholar ]
- Drolet MJ, Pinard C, Gaudet R. Les enjeux éthiques de la pratique privée: des ergothérapeutes du Québec lancent un cri d’alarme. Ethica – Revue interdisciplinaire de recherche en éthique. 2017; 21 (2):173–209. [ Google Scholar ]
- Drolet MJ, Ruest M. De l’éthique à l’ergothérapie: un cadre théorique et une méthode pour soutenir la pratique professionnelle. Québec: Presses de l’Université du Québec; 2021. [ Google Scholar ]
- Drolet, M. J., & Ruest, M. (accepted). Quels sont les enjeux éthiques soulevés par la recherche scientifique? In M. Lalancette & J. Luckerhoff (dir). Initiation au travail intellectuel et à la recherche . Québec: Presses de l’Université du Québec, 18 p
- Drolet MJ, Sauvageau A, Baril N, Gaudet R. Les enjeux éthiques de la formation clinique en ergothérapie. Revue Approches inductives. 2019; 6 (1):148–179. doi: 10.7202/1060048ar. [ CrossRef ] [ Google Scholar ]
- Fédération québécoise des professeures et des professeurs d’université (FQPPU) Enquête nationale sur la surcharge administrative du corps professoral universitaire québécois. Principaux résultats et pistes d’action. Montréal: FQPPU; 2019. [ Google Scholar ]
- Fortin MH. Fondements et étapes du processus de recherche. Méthodes quantitatives et qualitatives. Montréal, QC: Chenelière éducation; 2010. [ Google Scholar ]
- Fraser DM. Ethical dilemmas and practical problems for the practitioner researcher. Educational Action Research. 1997; 5 (1):161–171. doi: 10.1080/09650799700200014. [ CrossRef ] [ Google Scholar ]
- Fraser, N. (2011). Qu’est-ce que la justice sociale? Reconnaissance et redistribution . La Découverte
- Fricker, M. (2007). Epistemic Injustice: Power and the Ethics of Knowing . Oxford University Press
- Giorgi A, et al. De la méthode phénoménologique utilisée comme mode de recherche qualitative en sciences humaines: théories, pratique et évaluation. In: Poupart J, Groulx LH, Deslauriers JP, et al., editors. La recherche qualitative: enjeux épistémologiques et méthodologiques. Boucherville, QC: Gaëtan Morin; 1997. pp. 341–364. [ Google Scholar ]
- Giorgini V, Mecca JT, Gibson C, Medeiros K, Mumford MD, Connelly S, Devenport LD. Researcher Perceptions of Ethical Guidelines and Codes of Conduct. Accountability in Research. 2016; 22 (3):123–138. doi: 10.1080/08989621.2014.955607. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Glaser, J. W. (1994). Three realms of ethics: Individual, institutional, societal. Theoretical model and case studies . Kansas Cuty, Sheed & Ward
- Godrie B, Dos Santos M. Présentation: inégalités sociales, production des savoirs et de l’ignorance. Sociologie et sociétés. 2017; 49 (1):7. doi: 10.7202/1042804ar. [ CrossRef ] [ Google Scholar ]
- Hammell KW, Carpenter C, Dyck I. Using Qualitative Research: A Practical Introduction for Occupational and Physical Therapists. Edinburgh: Churchill Livingstone; 2000. [ Google Scholar ]
- Henderson M, Johnson NF, Auld G. Silences of ethical practice: dilemmas for researchers using social media. Educational Research and Evaluation. 2013; 19 (6):546–560. doi: 10.1080/13803611.2013.805656. [ CrossRef ] [ Google Scholar ]
- Husserl E. The crisis of European sciences and transcendental phenomenology. Evanston, IL: Northwestern University Press; 1970. [ Google Scholar ]
- Husserl E. The train of thoughts in the lectures. In: Polifroni EC, Welch M, editors. Perspectives on Philosophy of Science in Nursing. Philadelphia, PA: Lippincott; 1999. [ Google Scholar ]
- Hunt SD, Chonko LB, Wilcox JB. Ethical problems of marketing researchers. Journal of Marketing Research. 1984; 21 :309–324. doi: 10.1177/002224378402100308. [ CrossRef ] [ Google Scholar ]
- Hunt MR, Carnevale FA. Moral experience: A framework for bioethics research. Journal of Medical Ethics. 2011; 37 (11):658–662. doi: 10.1136/jme.2010.039008. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Jameton, A. (1984). Nursing practice: The ethical issues . Englewood Cliffs, Prentice-Hall
- Jarvis K. Dilemmas in International Research and the Value of Practical Wisdom. Developing World Bioethics. 2017; 17 (1):50–58. doi: 10.1111/dewb.12121. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Kahneman D. Système 1, système 2: les deux vitesses de la pensée. Paris: Flammarion; 2012. [ Google Scholar ]
- Keogh B, Daly L. The ethics of conducting research with mental health service users. British Journal of Nursing. 2009; 18 (5):277–281. doi: 10.12968/bjon.2009.18.5.40539. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Lierville AL, Grou C, Pelletier JF. Enjeux éthiques potentiels liés aux partenariats patients en psychiatrie: État de situation à l’Institut universitaire en santé mentale de Montréal. Santé mentale au Québec. 2015; 40 (1):119–134. doi: 10.7202/1032386ar. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Lynöe N, Sandlund M, Jacobsson L. Research ethics committees: A comparative study of assessment of ethical dilemmas. Scandinavian Journal of Public Health. 1999; 27 (2):152–159. doi: 10.1177/14034948990270020401. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Malherbe JF. Compromis, dilemmes et paradoxes en éthique clinique. Anjou: Éditions Fides; 1999. [ Google Scholar ]
- McGinn R. Discernment and denial: Nanotechnology researchers’ recognition of ethical responsibilities related to their work. NanoEthics. 2013; 7 :93–105. doi: 10.1007/s11569-013-0174-6. [ CrossRef ] [ Google Scholar ]
- Mills, C. W. (2017). Black Rights / White rongs. The Critique of Racial Liberalism . Oxford University Press
- Miyazaki AD, Taylor KA. Researcher interaction biases and business ethics research: Respondent reactions to researcher characteristics. Journal of Business Ethics. 2008; 81 (4):779–795. doi: 10.1007/s10551-007-9547-5. [ CrossRef ] [ Google Scholar ]
- Mondain N, Bologo E. L’intentionnalité du chercheur dans ses pratiques de production des connaissances: les enjeux soulevés par la construction des données en démographie et santé en Afrique. Cahiers de recherche sociologique. 2009; 48 :175–204. doi: 10.7202/039772ar. [ CrossRef ] [ Google Scholar ]
- Nicole, M., & Drolet, M. J. (in press). Fitting transphobia and cisgenderism in occupational therapy, Occupational Therapy Now
- Pope KS, Vetter VA. Ethical dilemmas encountered by members of the American Psychological Association: A national survey. The American Psychologist. 1992; 47 (3):397–411. doi: 10.1037/0003-066X.47.3.397. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Provencher V, Mortenson WB, Tanguay-Garneau L, Bélanger K, Dagenais M. Challenges and strategies pertaining to recruitment and retention of frail elderly in research studies: A systematic review. Archives of Gerontology and Geriatrics. 2014; 59 (1):18–24. doi: 10.1016/j.archger.2014.03.006. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Rawls, J. (1971). A Theory of Justice . Harvard University Press
- Resnik DB, Elliott KC. The Ethical Challenges of Socially Responsible Science. Accountability in Research. 2016; 23 (1):31–46. doi: 10.1080/08989621.2014.1002608. [ PMC free article ] [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Rosa, H. (2010). Accélération et aliénation. Vers une théorie critique de la modernité tardive . Paris, Découverte
- Sen, A. K. (2011). The Idea of Justice . The Belknap Press of Harvard University Press
- Sen, A. K. (1995). Inegality Reexaminated . Oxford University Press
- Sieber JE. Empirical Research on Research Ethics. Ethics & Behavior. 2004; 14 (4):397–412. doi: 10.1207/s15327019eb1404_9. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Sigmon ST. Ethical practices and beliefs of psychopathology researchers. Ethics & Behavior. 1995; 5 (4):295–309. doi: 10.1207/s15327019eb0504_1. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Swazey JP, Anderson MS, Lewis KS. Ethical Problems in Academic Research. American Scientist. 1993; 81 (6):542–553. [ Google Scholar ]
- Swisher LL, Arsalanian LE, Davis CM. The realm-individual-process-situation (RIPS) model of ethical decision-making. HPA Resource. 2005; 5 (3):3–8. [ Google Scholar ]
- Tri-Council Policy Statement (TCPS2) (2018). Ethical Conduct for Research Involving Humans . Government of Canada, Secretariat on Responsible Conduct of Research. https://ethics.gc.ca/eng/documents/tcps2-2018-en-interactive-final.pdf
- Thomas SP, Pollio HR. Listening to Patients: A Phenomenological Approach to Nursing Research and Practice. New York: Springer Publishing Company; 2002. [ Google Scholar ]
- Wiegand DL, Funk M. Consequences of clinical situations that cause critical care nurses to experience moral distress. Nursing Ethics. 2012; 19 (4):479–487. doi: 10.1177/0969733011429342. [ PubMed ] [ CrossRef ] [ Google Scholar ]
- Williams-Jones B, Potvin MJ, Mathieu G, Smith E. Barriers to research on research ethics review and conflicts of interest. IRB: Ethics & Human Research. 2013; 35 (5):14–20. [ PubMed ] [ Google Scholar ]
- Young, I. M. (2011). Justice and the Politics of difference . Princeton University Press
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Published: 13 September 2024
Research ethics matter
Nature Methods volume 21 , page 1569 ( 2024 ) Cite this article
Metrics details
- Biological sciences
- Biological techniques
- Institutions
- Research management
All life sciences research is potentially subject to ethical considerations. Institutions should support collaborations with professional ethicists and philosophers to help life scientists navigate ethical crossroads.
For scientists working with animals, human data or stem cells, or collecting ecological data in resource-poor settings, the ethical considerations of their research are something they must grapple with regularly. For those working at the molecular or cellular level or purely in silico, however, their experiences with ethics may be limited to a brief training course mainly covering issues of data manipulation and plagiarism.
Many molecular, cellular and computational biologists may think that research ethics — that is, whether the scientific questions being explored are ethical to pursue, whether the approaches used to pursue such questions are ethical, and whether the communication of that research is ethical — just do not apply to their work. But in this issue of Nature Methods , a Comment from Jeantine Lunshof and Julia Rijssenbeek 1 implores life scientists of all stripes to integrate discussions with professional ethicists and philosophers into the research planning process.
For some research fields, ethical guidelines are readily available. Research on human subjects — for example, the use of fMRI to study alterations to brain connectivity networks in disease — must be approved by ethics committees (in accordance with the Declaration of Helsinki ), and such subjects must provide informed consent. Experiments with lab mice must also be preapproved by an ethics committee, and researchers should follow the ARRIVE reporting guidelines 2 , as well as the American Veterinary Medical Association guidelines for animal euthanasia. Developmental biologists working with human embryos should abide by ethics guidelines set by the International Society for Stem Cell Research . Ecologists collecting data in the global South are encouraged to follow the TRUST Code recommendations — in particular, by including local researchers in the work.
There are many areas of life sciences research, however, where research does not require ethics oversight and ethics guidelines are not readily available, but where ethical considerations may yet be important. There are likely several examples of methodologies and tools that, in the wrong hands, may lead to harmful gain-of-function experiments or dual-use threats to public health or national security. A molecular biologist tinkering to improve genome editing tools would be wise to ponder the implications of the unethical use of the technology for human germline editing, as in the ‘CRISPR babies’ case 3 . Computational biologists using AI technology for drug discovery need to be aware that such models could be misused to design biochemical weapons 4 .
In their Comment, Lunshof and Rijssenbeek describe a model called ‘collaborative ethics’, which calls for life sciences researchers to work closely with professional ethicists or philosophers starting at the earliest stages of research planning. As they write, collaboration can “improve the efficiency and robustness of outcomes” for the research team and also “prepare a team for a formal ethics review and criticism after publication.” Such collaborations also benefit the field of philosophy, “as direct involvement with the sciences allows long-held assumptions and arguments to be put to the test.”
In the collaborative ethics model, a research team will discuss their ideas and concepts with a professional ethicist or philosopher, who may ask probing questions about the nature of the work. The ethicist will help the researchers understand whether there are any ethical considerations and whether the potential harms of sharing the resulting new knowledge with society outweigh the potential benefits of advancing science. Lunshof and Rijssenbeek highlight how the collaborative ethics model played a role in three different projects: the engineering of synthetic human entities with embryo-like features, the development of brain organoids and the programming of computer-designed ‘biobots’. They note that collaborative ethics may also have a role to play in many other fields, such as protein engineering, systems biology, aging research and computational biology.
As described in our journal policy , performing ethical research is the responsibility of all scientists. Professional ethicists are not the police; their role is not to punish or curb scientific progress. Rather, ethicists can serve as a ‘conscience’ and help scientists understand the ethical implications of their work. Without such collaborations, the after-the-fact consequences could be much worse — yes, papers can be retracted, but reputations are already damaged and harmful gain-of-function work may be out there for the world to see.
For scientists used to dealing with quantitative measures and statistical significance, the concept of research ethics can feel a bit vague or murky, at best an afterthought to their work. This is even more a reason for breaking down the walls between science and ethics, especially in this age of rapid technology development and especially with swift advances in AI. We strongly encourage institutions to go beyond minimal ethics training courses and do much more to support close collaborations between scientists and professional ethicists.
Lunshof, J. E. & Rijssenbeek, J. https://doi.org/10.1038/s41592-024-02320-8 (2024).
Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. & Altman, D. G. PLoS Biol. 8 , e1000412 (2010).
Article PubMed PubMed Central Google Scholar
Cyranoski, D. & Ledford, H. Nature 563 , 607–608 (2018).
Article CAS PubMed Google Scholar
Urbina, F., Lentzos, F., Invernizzi, C. & Ekins, S. Nat. Mach. Intell. 4 , 189–191 (2022).
Download references
Rights and permissions
Reprints and permissions
About this article
Cite this article.
Research ethics matter. Nat Methods 21 , 1569 (2024). https://doi.org/10.1038/s41592-024-02425-0
Download citation
Published : 13 September 2024
Issue Date : September 2024
DOI : https://doi.org/10.1038/s41592-024-02425-0
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.
Peer Reviewed
GPT-fabricated scientific papers on Google Scholar: Key features, spread, and implications for preempting evidence manipulation
Article metrics.
CrossRef Citations
Altmetric Score
PDF Downloads
Academic journals, archives, and repositories are seeing an increasing number of questionable research papers clearly produced using generative AI. They are often created with widely available, general-purpose AI applications, most likely ChatGPT, and mimic scientific writing. Google Scholar easily locates and lists these questionable papers alongside reputable, quality-controlled research. Our analysis of a selection of questionable GPT-fabricated scientific papers found in Google Scholar shows that many are about applied, often controversial topics susceptible to disinformation: the environment, health, and computing. The resulting enhanced potential for malicious manipulation of society’s evidence base, particularly in politically divisive domains, is a growing concern.
Swedish School of Library and Information Science, University of Borås, Sweden
Department of Arts and Cultural Sciences, Lund University, Sweden
Division of Environmental Communication, Swedish University of Agricultural Sciences, Sweden

Research Questions
- Where are questionable publications produced with generative pre-trained transformers (GPTs) that can be found via Google Scholar published or deposited?
- What are the main characteristics of these publications in relation to predominant subject categories?
- How are these publications spread in the research infrastructure for scholarly communication?
- How is the role of the scholarly communication infrastructure challenged in maintaining public trust in science and evidence through inappropriate use of generative AI?
research note Summary
- A sample of scientific papers with signs of GPT-use found on Google Scholar was retrieved, downloaded, and analyzed using a combination of qualitative coding and descriptive statistics. All papers contained at least one of two common phrases returned by conversational agents that use large language models (LLM) like OpenAI’s ChatGPT. Google Search was then used to determine the extent to which copies of questionable, GPT-fabricated papers were available in various repositories, archives, citation databases, and social media platforms.
- Roughly two-thirds of the retrieved papers were found to have been produced, at least in part, through undisclosed, potentially deceptive use of GPT. The majority (57%) of these questionable papers dealt with policy-relevant subjects (i.e., environment, health, computing), susceptible to influence operations. Most were available in several copies on different domains (e.g., social media, archives, and repositories).
- Two main risks arise from the increasingly common use of GPT to (mass-)produce fake, scientific publications. First, the abundance of fabricated “studies” seeping into all areas of the research infrastructure threatens to overwhelm the scholarly communication system and jeopardize the integrity of the scientific record. A second risk lies in the increased possibility that convincingly scientific-looking content was in fact deceitfully created with AI tools and is also optimized to be retrieved by publicly available academic search engines, particularly Google Scholar. However small, this possibility and awareness of it risks undermining the basis for trust in scientific knowledge and poses serious societal risks.
Implications
The use of ChatGPT to generate text for academic papers has raised concerns about research integrity. Discussion of this phenomenon is ongoing in editorials, commentaries, opinion pieces, and on social media (Bom, 2023; Stokel-Walker, 2024; Thorp, 2023). There are now several lists of papers suspected of GPT misuse, and new papers are constantly being added. 1 See for example Academ-AI, https://www.academ-ai.info/ , and Retraction Watch, https://retractionwatch.com/papers-and-peer-reviews-with-evidence-of-chatgpt-writing/ . While many legitimate uses of GPT for research and academic writing exist (Huang & Tan, 2023; Kitamura, 2023; Lund et al., 2023), its undeclared use—beyond proofreading—has potentially far-reaching implications for both science and society, but especially for their relationship. It, therefore, seems important to extend the discussion to one of the most accessible and well-known intermediaries between science, but also certain types of misinformation, and the public, namely Google Scholar, also in response to the legitimate concerns that the discussion of generative AI and misinformation needs to be more nuanced and empirically substantiated (Simon et al., 2023).
Google Scholar, https://scholar.google.com , is an easy-to-use academic search engine. It is available for free, and its index is extensive (Gusenbauer & Haddaway, 2020). It is also often touted as a credible source for academic literature and even recommended in library guides, by media and information literacy initiatives, and fact checkers (Tripodi et al., 2023). However, Google Scholar lacks the transparency and adherence to standards that usually characterize citation databases. Instead, Google Scholar uses automated crawlers, like Google’s web search engine (Martín-Martín et al., 2021), and the inclusion criteria are based on primarily technical standards, allowing any individual author—with or without scientific affiliation—to upload papers to be indexed (Google Scholar Help, n.d.). It has been shown that Google Scholar is susceptible to manipulation through citation exploits (Antkare, 2020) and by providing access to fake scientific papers (Dadkhah et al., 2017). A large part of Google Scholar’s index consists of publications from established scientific journals or other forms of quality-controlled, scholarly literature. However, the index also contains a large amount of gray literature, including student papers, working papers, reports, preprint servers, and academic networking sites, as well as material from so-called “questionable” academic journals, including paper mills. The search interface does not offer the possibility to filter the results meaningfully by material type, publication status, or form of quality control, such as limiting the search to peer-reviewed material.
To understand the occurrence of ChatGPT (co-)authored work in Google Scholar’s index, we scraped it for publications, including one of two common ChatGPT responses (see Appendix A) that we encountered on social media and in media reports (DeGeurin, 2024). The results of our descriptive statistical analyses showed that around 62% did not declare the use of GPTs. Most of these GPT-fabricated papers were found in non-indexed journals and working papers, but some cases included research published in mainstream scientific journals and conference proceedings. 2 Indexed journals mean scholarly journals indexed by abstract and citation databases such as Scopus and Web of Science, where the indexation implies journals with high scientific quality. Non-indexed journals are journals that fall outside of this indexation. More than half (57%) of these GPT-fabricated papers concerned policy-relevant subject areas susceptible to influence operations. To avoid increasing the visibility of these publications, we abstained from referencing them in this research note. However, we have made the data available in the Harvard Dataverse repository.
The publications were related to three issue areas—health (14.5%), environment (19.5%) and computing (23%)—with key terms such “healthcare,” “COVID-19,” or “infection”for health-related papers, and “analysis,” “sustainable,” and “global” for environment-related papers. In several cases, the papers had titles that strung together general keywords and buzzwords, thus alluding to very broad and current research. These terms included “biology,” “telehealth,” “climate policy,” “diversity,” and “disrupting,” to name just a few. While the study’s scope and design did not include a detailed analysis of which parts of the articles included fabricated text, our dataset did contain the surrounding sentences for each occurrence of the suspicious phrases that formed the basis for our search and subsequent selection. Based on that, we can say that the phrases occurred in most sections typically found in scientific publications, including the literature review, methods, conceptual and theoretical frameworks, background, motivation or societal relevance, and even discussion. This was confirmed during the joint coding, where we read and discussed all articles. It became clear that not just the text related to the telltale phrases was created by GPT, but that almost all articles in our sample of questionable articles likely contained traces of GPT-fabricated text everywhere.
Evidence hacking and backfiring effects
Generative pre-trained transformers (GPTs) can be used to produce texts that mimic scientific writing. These texts, when made available online—as we demonstrate—leak into the databases of academic search engines and other parts of the research infrastructure for scholarly communication. This development exacerbates problems that were already present with less sophisticated text generators (Antkare, 2020; Cabanac & Labbé, 2021). Yet, the public release of ChatGPT in 2022, together with the way Google Scholar works, has increased the likelihood of lay people (e.g., media, politicians, patients, students) coming across questionable (or even entirely GPT-fabricated) papers and other problematic research findings. Previous research has emphasized that the ability to determine the value and status of scientific publications for lay people is at stake when misleading articles are passed off as reputable (Haider & Åström, 2017) and that systematic literature reviews risk being compromised (Dadkhah et al., 2017). It has also been highlighted that Google Scholar, in particular, can be and has been exploited for manipulating the evidence base for politically charged issues and to fuel conspiracy narratives (Tripodi et al., 2023). Both concerns are likely to be magnified in the future, increasing the risk of what we suggest calling evidence hacking —the strategic and coordinated malicious manipulation of society’s evidence base.
The authority of quality-controlled research as evidence to support legislation, policy, politics, and other forms of decision-making is undermined by the presence of undeclared GPT-fabricated content in publications professing to be scientific. Due to the large number of archives, repositories, mirror sites, and shadow libraries to which they spread, there is a clear risk that GPT-fabricated, questionable papers will reach audiences even after a possible retraction. There are considerable technical difficulties involved in identifying and tracing computer-fabricated papers (Cabanac & Labbé, 2021; Dadkhah et al., 2023; Jones, 2024), not to mention preventing and curbing their spread and uptake.
However, as the rise of the so-called anti-vaxx movement during the COVID-19 pandemic and the ongoing obstruction and denial of climate change show, retracting erroneous publications often fuels conspiracies and increases the following of these movements rather than stopping them. To illustrate this mechanism, climate deniers frequently question established scientific consensus by pointing to other, supposedly scientific, studies that support their claims. Usually, these are poorly executed, not peer-reviewed, based on obsolete data, or even fraudulent (Dunlap & Brulle, 2020). A similar strategy is successful in the alternative epistemic world of the global anti-vaccination movement (Carrion, 2018) and the persistence of flawed and questionable publications in the scientific record already poses significant problems for health research, policy, and lawmakers, and thus for society as a whole (Littell et al., 2024). Considering that a person’s support for “doing your own research” is associated with increased mistrust in scientific institutions (Chinn & Hasell, 2023), it will be of utmost importance to anticipate and consider such backfiring effects already when designing a technical solution, when suggesting industry or legal regulation, and in the planning of educational measures.
Recommendations
Solutions should be based on simultaneous considerations of technical, educational, and regulatory approaches, as well as incentives, including social ones, across the entire research infrastructure. Paying attention to how these approaches and incentives relate to each other can help identify points and mechanisms for disruption. Recognizing fraudulent academic papers must happen alongside understanding how they reach their audiences and what reasons there might be for some of these papers successfully “sticking around.” A possible way to mitigate some of the risks associated with GPT-fabricated scholarly texts finding their way into academic search engine results would be to provide filtering options for facets such as indexed journals, gray literature, peer-review, and similar on the interface of publicly available academic search engines. Furthermore, evaluation tools for indexed journals 3 Such as LiU Journal CheckUp, https://ep.liu.se/JournalCheckup/default.aspx?lang=eng . could be integrated into the graphical user interfaces and the crawlers of these academic search engines. To enable accountability, it is important that the index (database) of such a search engine is populated according to criteria that are transparent, open to scrutiny, and appropriate to the workings of science and other forms of academic research. Moreover, considering that Google Scholar has no real competitor, there is a strong case for establishing a freely accessible, non-specialized academic search engine that is not run for commercial reasons but for reasons of public interest. Such measures, together with educational initiatives aimed particularly at policymakers, science communicators, journalists, and other media workers, will be crucial to reducing the possibilities for and effects of malicious manipulation or evidence hacking. It is important not to present this as a technical problem that exists only because of AI text generators but to relate it to the wider concerns in which it is embedded. These range from a largely dysfunctional scholarly publishing system (Haider & Åström, 2017) and academia’s “publish or perish” paradigm to Google’s near-monopoly and ideological battles over the control of information and ultimately knowledge. Any intervention is likely to have systemic effects; these effects need to be considered and assessed in advance and, ideally, followed up on.
Our study focused on a selection of papers that were easily recognizable as fraudulent. We used this relatively small sample as a magnifying glass to examine, delineate, and understand a problem that goes beyond the scope of the sample itself, which however points towards larger concerns that require further investigation. The work of ongoing whistleblowing initiatives 4 Such as Academ-AI, https://www.academ-ai.info/ , and Retraction Watch, https://retractionwatch.com/papers-and-peer-reviews-with-evidence-of-chatgpt-writing/ . , recent media reports of journal closures (Subbaraman, 2024), or GPT-related changes in word use and writing style (Cabanac et al., 2021; Stokel-Walker, 2024) suggest that we only see the tip of the iceberg. There are already more sophisticated cases (Dadkhah et al., 2023) as well as cases involving fabricated images (Gu et al., 2022). Our analysis shows that questionable and potentially manipulative GPT-fabricated papers permeate the research infrastructure and are likely to become a widespread phenomenon. Our findings underline that the risk of fake scientific papers being used to maliciously manipulate evidence (see Dadkhah et al., 2017) must be taken seriously. Manipulation may involve undeclared automatic summaries of texts, inclusion in literature reviews, explicit scientific claims, or the concealment of errors in studies so that they are difficult to detect in peer review. However, the mere possibility of these things happening is a significant risk in its own right that can be strategically exploited and will have ramifications for trust in and perception of science. Society’s methods of evaluating sources and the foundations of media and information literacy are under threat and public trust in science is at risk of further erosion, with far-reaching consequences for society in dealing with information disorders. To address this multifaceted problem, we first need to understand why it exists and proliferates.
Finding 1: 139 GPT-fabricated, questionable papers were found and listed as regular results on the Google Scholar results page. Non-indexed journals dominate.
Most questionable papers we found were in non-indexed journals or were working papers, but we did also find some in established journals, publications, conferences, and repositories. We found a total of 139 papers with a suspected deceptive use of ChatGPT or similar LLM applications (see Table 1). Out of these, 19 were in indexed journals, 89 were in non-indexed journals, 19 were student papers found in university databases, and 12 were working papers (mostly in preprint databases). Table 1 divides these papers into categories. Health and environment papers made up around 34% (47) of the sample. Of these, 66% were present in non-indexed journals.
| Indexed journals* | 5 | 3 | 4 | 7 | 19 |
| Non-indexed journals | 18 | 18 | 13 | 40 | 89 |
| Student papers | 4 | 3 | 1 | 11 | 19 |
| Working papers | 5 | 3 | 2 | 2 | 12 |
| Total | 32 | 27 | 20 | 60 | 139 |
Finding 2: GPT-fabricated, questionable papers are disseminated online, permeating the research infrastructure for scholarly communication, often in multiple copies. Applied topics with practical implications dominate.
The 20 papers concerning health-related issues are distributed across 20 unique domains, accounting for 46 URLs. The 27 papers dealing with environmental issues can be found across 26 unique domains, accounting for 56 URLs. Most of the identified papers exist in multiple copies and have already spread to several archives, repositories, and social media. It would be difficult, or impossible, to remove them from the scientific record.
As apparent from Table 2, GPT-fabricated, questionable papers are seeping into most parts of the online research infrastructure for scholarly communication. Platforms on which identified papers have appeared include ResearchGate, ORCiD, Journal of Population Therapeutics and Clinical Pharmacology (JPTCP), Easychair, Frontiers, the Institute of Electrical and Electronics Engineer (IEEE), and X/Twitter. Thus, even if they are retracted from their original source, it will prove very difficult to track, remove, or even just mark them up on other platforms. Moreover, unless regulated, Google Scholar will enable their continued and most likely unlabeled discoverability.
| Environment | researchgate.net (13) | orcid.org (4) | easychair.org (3) | ijope.com* (3) | publikasiindonesia.id (3) |
| Health | researchgate.net (15) | ieee.org (4) | twitter.com (3) | jptcp.com** (2) | frontiersin.org (2) |
A word rain visualization (Centre for Digital Humanities Uppsala, 2023), which combines word prominences through TF-IDF 5 Term frequency–inverse document frequency , a method for measuring the significance of a word in a document compared to its frequency across all documents in a collection. scores with semantic similarity of the full texts of our sample of GPT-generated articles that fall into the “Environment” and “Health” categories, reflects the two categories in question. However, as can be seen in Figure 1, it also reveals overlap and sub-areas. The y-axis shows word prominences through word positions and font sizes, while the x-axis indicates semantic similarity. In addition to a certain amount of overlap, this reveals sub-areas, which are best described as two distinct events within the word rain. The event on the left bundles terms related to the development and management of health and healthcare with “challenges,” “impact,” and “potential of artificial intelligence”emerging as semantically related terms. Terms related to research infrastructures, environmental, epistemic, and technological concepts are arranged further down in the same event (e.g., “system,” “climate,” “understanding,” “knowledge,” “learning,” “education,” “sustainable”). A second distinct event further to the right bundles terms associated with fish farming and aquatic medicinal plants, highlighting the presence of an aquaculture cluster. Here, the prominence of groups of terms such as “used,” “model,” “-based,” and “traditional” suggests the presence of applied research on these topics. The two events making up the word rain visualization, are linked by a less dominant but overlapping cluster of terms related to “energy” and “water.”
The bar chart of the terms in the paper subset (see Figure 2) complements the word rain visualization by depicting the most prominent terms in the full texts along the y-axis. Here, word prominences across health and environment papers are arranged descendingly, where values outside parentheses are TF-IDF values (relative frequencies) and values inside parentheses are raw term frequencies (absolute frequencies).
Finding 3: Google Scholar presents results from quality-controlled and non-controlled citation databases on the same interface, providing unfiltered access to GPT-fabricated questionable papers.
Google Scholar’s central position in the publicly accessible scholarly communication infrastructure, as well as its lack of standards, transparency, and accountability in terms of inclusion criteria, has potentially serious implications for public trust in science. This is likely to exacerbate the already-known potential to exploit Google Scholar for evidence hacking (Tripodi et al., 2023) and will have implications for any attempts to retract or remove fraudulent papers from their original publication venues. Any solution must consider the entirety of the research infrastructure for scholarly communication and the interplay of different actors, interests, and incentives.
We searched and scraped Google Scholar using the Python library Scholarly (Cholewiak et al., 2023) for papers that included specific phrases known to be common responses from ChatGPT and similar applications with the same underlying model (GPT3.5 or GPT4): “as of my last knowledge update” and/or “I don’t have access to real-time data” (see Appendix A). This facilitated the identification of papers that likely used generative AI to produce text, resulting in 227 retrieved papers. The papers’ bibliographic information was automatically added to a spreadsheet and downloaded into Zotero. 6 An open-source reference manager, https://zotero.org .
We employed multiple coding (Barbour, 2001) to classify the papers based on their content. First, we jointly assessed whether the paper was suspected of fraudulent use of ChatGPT (or similar) based on how the text was integrated into the papers and whether the paper was presented as original research output or the AI tool’s role was acknowledged. Second, in analyzing the content of the papers, we continued the multiple coding by classifying the fraudulent papers into four categories identified during an initial round of analysis—health, environment, computing, and others—and then determining which subjects were most affected by this issue (see Table 1). Out of the 227 retrieved papers, 88 papers were written with legitimate and/or declared use of GPTs (i.e., false positives, which were excluded from further analysis), and 139 papers were written with undeclared and/or fraudulent use (i.e., true positives, which were included in further analysis). The multiple coding was conducted jointly by all authors of the present article, who collaboratively coded and cross-checked each other’s interpretation of the data simultaneously in a shared spreadsheet file. This was done to single out coding discrepancies and settle coding disagreements, which in turn ensured methodological thoroughness and analytical consensus (see Barbour, 2001). Redoing the category coding later based on our established coding schedule, we achieved an intercoder reliability (Cohen’s kappa) of 0.806 after eradicating obvious differences.
The ranking algorithm of Google Scholar prioritizes highly cited and older publications (Martín-Martín et al., 2016). Therefore, the position of the articles on the search engine results pages was not particularly informative, considering the relatively small number of results in combination with the recency of the publications. Only the query “as of my last knowledge update” had more than two search engine result pages. On those, questionable articles with undeclared use of GPTs were evenly distributed across all result pages (min: 4, max: 9, mode: 8), with the proportion of undeclared use being slightly higher on average on later search result pages.
To understand how the papers making fraudulent use of generative AI were disseminated online, we programmatically searched for the paper titles (with exact string matching) in Google Search from our local IP address (see Appendix B) using the googlesearch – python library(Vikramaditya, 2020). We manually verified each search result to filter out false positives—results that were not related to the paper—and then compiled the most prominent URLs by field. This enabled the identification of other platforms through which the papers had been spread. We did not, however, investigate whether copies had spread into SciHub or other shadow libraries, or if they were referenced in Wikipedia.
We used descriptive statistics to count the prevalence of the number of GPT-fabricated papers across topics and venues and top domains by subject. The pandas software library for the Python programming language (The pandas development team, 2024) was used for this part of the analysis. Based on the multiple coding, paper occurrences were counted in relation to their categories, divided into indexed journals, non-indexed journals, student papers, and working papers. The schemes, subdomains, and subdirectories of the URL strings were filtered out while top-level domains and second-level domains were kept, which led to normalizing domain names. This, in turn, allowed the counting of domain frequencies in the environment and health categories. To distinguish word prominences and meanings in the environment and health-related GPT-fabricated questionable papers, a semantically-aware word cloud visualization was produced through the use of a word rain (Centre for Digital Humanities Uppsala, 2023) for full-text versions of the papers. Font size and y-axis positions indicate word prominences through TF-IDF scores for the environment and health papers (also visualized in a separate bar chart with raw term frequencies in parentheses), and words are positioned along the x-axis to reflect semantic similarity (Skeppstedt et al., 2024), with an English Word2vec skip gram model space (Fares et al., 2017). An English stop word list was used, along with a manually produced list including terms such as “https,” “volume,” or “years.”
- Artificial Intelligence
- / Search engines
Cite this Essay
Haider, J., Söderström, K. R., Ekström, B., & Rödl, M. (2024). GPT-fabricated scientific papers on Google Scholar: Key features, spread, and implications for preempting evidence manipulation. Harvard Kennedy School (HKS) Misinformation Review . https://doi.org/10.37016/mr-2020-156
- / Appendix B
Bibliography
Antkare, I. (2020). Ike Antkare, his publications, and those of his disciples. In M. Biagioli & A. Lippman (Eds.), Gaming the metrics (pp. 177–200). The MIT Press. https://doi.org/10.7551/mitpress/11087.003.0018
Barbour, R. S. (2001). Checklists for improving rigour in qualitative research: A case of the tail wagging the dog? BMJ , 322 (7294), 1115–1117. https://doi.org/10.1136/bmj.322.7294.1115
Bom, H.-S. H. (2023). Exploring the opportunities and challenges of ChatGPT in academic writing: A roundtable discussion. Nuclear Medicine and Molecular Imaging , 57 (4), 165–167. https://doi.org/10.1007/s13139-023-00809-2
Cabanac, G., & Labbé, C. (2021). Prevalence of nonsensical algorithmically generated papers in the scientific literature. Journal of the Association for Information Science and Technology , 72 (12), 1461–1476. https://doi.org/10.1002/asi.24495
Cabanac, G., Labbé, C., & Magazinov, A. (2021). Tortured phrases: A dubious writing style emerging in science. Evidence of critical issues affecting established journals . arXiv. https://doi.org/10.48550/arXiv.2107.06751
Carrion, M. L. (2018). “You need to do your research”: Vaccines, contestable science, and maternal epistemology. Public Understanding of Science , 27 (3), 310–324. https://doi.org/10.1177/0963662517728024
Centre for Digital Humanities Uppsala (2023). CDHUppsala/word-rain [Computer software]. https://github.com/CDHUppsala/word-rain
Chinn, S., & Hasell, A. (2023). Support for “doing your own research” is associated with COVID-19 misperceptions and scientific mistrust. Harvard Kennedy School (HSK) Misinformation Review, 4 (3). https://doi.org/10.37016/mr-2020-117
Cholewiak, S. A., Ipeirotis, P., Silva, V., & Kannawadi, A. (2023). SCHOLARLY: Simple access to Google Scholar authors and citation using Python (1.5.0) [Computer software]. https://doi.org/10.5281/zenodo.5764801
Dadkhah, M., Lagzian, M., & Borchardt, G. (2017). Questionable papers in citation databases as an issue for literature review. Journal of Cell Communication and Signaling , 11 (2), 181–185. https://doi.org/10.1007/s12079-016-0370-6
Dadkhah, M., Oermann, M. H., Hegedüs, M., Raman, R., & Dávid, L. D. (2023). Detection of fake papers in the era of artificial intelligence. Diagnosis , 10 (4), 390–397. https://doi.org/10.1515/dx-2023-0090
DeGeurin, M. (2024, March 19). AI-generated nonsense is leaking into scientific journals. Popular Science. https://www.popsci.com/technology/ai-generated-text-scientific-journals/
Dunlap, R. E., & Brulle, R. J. (2020). Sources and amplifiers of climate change denial. In D.C. Holmes & L. M. Richardson (Eds.), Research handbook on communicating climate change (pp. 49–61). Edward Elgar Publishing. https://doi.org/10.4337/9781789900408.00013
Fares, M., Kutuzov, A., Oepen, S., & Velldal, E. (2017). Word vectors, reuse, and replicability: Towards a community repository of large-text resources. In J. Tiedemann & N. Tahmasebi (Eds.), Proceedings of the 21st Nordic Conference on Computational Linguistics (pp. 271–276). Association for Computational Linguistics. https://aclanthology.org/W17-0237
Google Scholar Help. (n.d.). Inclusion guidelines for webmasters . https://scholar.google.com/intl/en/scholar/inclusion.html
Gu, J., Wang, X., Li, C., Zhao, J., Fu, W., Liang, G., & Qiu, J. (2022). AI-enabled image fraud in scientific publications. Patterns , 3 (7), 100511. https://doi.org/10.1016/j.patter.2022.100511
Gusenbauer, M., & Haddaway, N. R. (2020). Which academic search systems are suitable for systematic reviews or meta-analyses? Evaluating retrieval qualities of Google Scholar, PubMed, and 26 other resources. Research Synthesis Methods , 11 (2), 181–217. https://doi.org/10.1002/jrsm.1378
Haider, J., & Åström, F. (2017). Dimensions of trust in scholarly communication: Problematizing peer review in the aftermath of John Bohannon’s “Sting” in science. Journal of the Association for Information Science and Technology , 68 (2), 450–467. https://doi.org/10.1002/asi.23669
Huang, J., & Tan, M. (2023). The role of ChatGPT in scientific communication: Writing better scientific review articles. American Journal of Cancer Research , 13 (4), 1148–1154. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10164801/
Jones, N. (2024). How journals are fighting back against a wave of questionable images. Nature , 626 (8000), 697–698. https://doi.org/10.1038/d41586-024-00372-6
Kitamura, F. C. (2023). ChatGPT is shaping the future of medical writing but still requires human judgment. Radiology , 307 (2), e230171. https://doi.org/10.1148/radiol.230171
Littell, J. H., Abel, K. M., Biggs, M. A., Blum, R. W., Foster, D. G., Haddad, L. B., Major, B., Munk-Olsen, T., Polis, C. B., Robinson, G. E., Rocca, C. H., Russo, N. F., Steinberg, J. R., Stewart, D. E., Stotland, N. L., Upadhyay, U. D., & Ditzhuijzen, J. van. (2024). Correcting the scientific record on abortion and mental health outcomes. BMJ , 384 , e076518. https://doi.org/10.1136/bmj-2023-076518
Lund, B. D., Wang, T., Mannuru, N. R., Nie, B., Shimray, S., & Wang, Z. (2023). ChatGPT and a new academic reality: Artificial Intelligence-written research papers and the ethics of the large language models in scholarly publishing. Journal of the Association for Information Science and Technology, 74 (5), 570–581. https://doi.org/10.1002/asi.24750
Martín-Martín, A., Orduna-Malea, E., Ayllón, J. M., & Delgado López-Cózar, E. (2016). Back to the past: On the shoulders of an academic search engine giant. Scientometrics , 107 , 1477–1487. https://doi.org/10.1007/s11192-016-1917-2
Martín-Martín, A., Thelwall, M., Orduna-Malea, E., & Delgado López-Cózar, E. (2021). Google Scholar, Microsoft Academic, Scopus, Dimensions, Web of Science, and OpenCitations’ COCI: A multidisciplinary comparison of coverage via citations. Scientometrics , 126 (1), 871–906. https://doi.org/10.1007/s11192-020-03690-4
Simon, F. M., Altay, S., & Mercier, H. (2023). Misinformation reloaded? Fears about the impact of generative AI on misinformation are overblown. Harvard Kennedy School (HKS) Misinformation Review, 4 (5). https://doi.org/10.37016/mr-2020-127
Skeppstedt, M., Ahltorp, M., Kucher, K., & Lindström, M. (2024). From word clouds to Word Rain: Revisiting the classic word cloud to visualize climate change texts. Information Visualization , 23 (3), 217–238. https://doi.org/10.1177/14738716241236188
Swedish Research Council. (2017). Good research practice. Vetenskapsrådet.
Stokel-Walker, C. (2024, May 1.). AI Chatbots Have Thoroughly Infiltrated Scientific Publishing . Scientific American. https://www.scientificamerican.com/article/chatbots-have-thoroughly-infiltrated-scientific-publishing/
Subbaraman, N. (2024, May 14). Flood of fake science forces multiple journal closures: Wiley to shutter 19 more journals, some tainted by fraud. The Wall Street Journal . https://www.wsj.com/science/academic-studies-research-paper-mills-journals-publishing-f5a3d4bc
The pandas development team. (2024). pandas-dev/pandas: Pandas (v2.2.2) [Computer software]. Zenodo. https://doi.org/10.5281/zenodo.10957263
Thorp, H. H. (2023). ChatGPT is fun, but not an author. Science , 379 (6630), 313–313. https://doi.org/10.1126/science.adg7879
Tripodi, F. B., Garcia, L. C., & Marwick, A. E. (2023). ‘Do your own research’: Affordance activation and disinformation spread. Information, Communication & Society , 27 (6), 1212–1228. https://doi.org/10.1080/1369118X.2023.2245869
Vikramaditya, N. (2020). Nv7-GitHub/googlesearch [Computer software]. https://github.com/Nv7-GitHub/googlesearch
This research has been supported by Mistra, the Swedish Foundation for Strategic Environmental Research, through the research program Mistra Environmental Communication (Haider, Ekström, Rödl) and the Marcus and Amalia Wallenberg Foundation [2020.0004] (Söderström).
Competing Interests
The authors declare no competing interests.
The research described in this article was carried out under Swedish legislation. According to the relevant EU and Swedish legislation (2003:460) on the ethical review of research involving humans (“Ethical Review Act”), the research reported on here is not subject to authorization by the Swedish Ethical Review Authority (“etikprövningsmyndigheten”) (SRC, 2017).
This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and reproduction in any medium, provided that the original author and source are properly credited.
Data Availability
All data needed to replicate this study are available at the Harvard Dataverse: https://doi.org/10.7910/DVN/WUVD8X
Acknowledgements
The authors wish to thank two anonymous reviewers for their valuable comments on the article manuscript as well as the editorial group of Harvard Kennedy School (HKS) Misinformation Review for their thoughtful feedback and input.

IMAGES
VIDEO
COMMENTS
The ideas and opinions expressed in this essay are the author's own and do not necessarily represent those of the NIH, NIEHS, or US government. ... Education in research ethics is can help people get a better understanding of ethical standards, policies, and issues and improve ethical judgment and decision making. ...
Ethical considerations in research are a set of principles that guide your research designs and practices. Scientists and researchers must always adhere to a certain code of conduct when collecting data from people. The goals of human research often include understanding real-life phenomena, studying effective treatments, investigating ...
business. Ethical norms also serve the aims or goals of research and apply to people who conduct scientific research or other scholarly or creative activities. There is even a specialized discipline, research ethics, which studies these norms. There are several reasons why it is important to adhere to ethical norms in research. First, norms ...
Essay. Print PDF. Description. Research Ethics is defined here to be the ethics of the planning, conduct, and reporting of research. This introduction covers what research ethics is, its ethical distinctions, approaches to teaching research ethics, and other resources on this topic.
Ethics Principle 1: Respect for persons. As the name suggests, this principle is all about ensuring that your participants are treated fairly and respectfully. In practical terms, this means informed consent - in other words, participants should be fully informed about the nature of the research, as well as any potential risks. Additionally ...
Ethics are a guiding principle that shapes the conduct of researchers. It influences both the process of discovery and the implications and applications of scientific findings 1. Ethical considerations in research include, but are not limited to, the management of data, the responsible use of resources, respect for human rights, the treatment ...
Ethics in research, writing, and publication are critical in medicine and nursing—decisions that affect human lives often are influenced by knowledge that is disseminated in healthcare journals. While it may seem less critical that healthcare design adhere to strong ethical principles in research, writing, and publication of findings, huge ...
Revised on 6 July 2024. Ethical considerations in research are a set of principles that guide your research designs and practices. Scientists and researchers must always adhere to a certain code of conduct when collecting data from people. The goals of human research often include understanding real-life phenomena, studying effective treatments ...
A checklist to ensure you're ethics-check ready. Before you begin your research. Obtain approval from your IRB, IACUC or other approving body. Obtain written informed consent from human participants, guardians or next-of-kin. Obtain permits or permission from property owners, or confirm that permits are not required.
Abstract. Ethics, as an integral component of human decision-making, undeniably shape the landscape of scientific research. This article delves deeply into the nuanced realm of ethical ...
Ethics and ethical behaviour are the fundamental pillars of a civilised society. The focus on ethical behaviour is indispensable in certain fields such as medicine, finance, or law. In fact, ethics gets precedence with anything that would include, affect, transform, or influence upon individuals, communities or any living creatures. Many institutions within Europe have set up their own ...
Research Ethics is defined here to be the ethics of the planning, conduct, and reporting of research. It is clear that research ethics should include: Protections of human and animal subjects; However, not all researchers use human or animal subjects, nor are the ethical dimensions of research confined solely to protections for research subjects.
Introduction. In science and medical research, ethics is essential in enhancing the safety and well-being of the subjects or participants. Different studies globally expose vulnerable populations or subjects to abuse, affecting their overall health. In the same case, researchers are employing diverse strategies to enhance ethics and reduce ...
What is Ethics in Research and Why is it Important? This article defines ethics in research, including codes and policies, and demonstrates the importance of ethics in research. The School of Medicine acknowledges and honors that the University stands on the unceded land of the Coast Salish peoples, land which touches the shared waters of all ...
Ethical Considerations. Ethical considerations in research refer to the principles and guidelines that researchers must follow to ensure that their studies are conducted in an ethical and responsible manner. These considerations are designed to protect the rights, safety, and well-being of research participants, as well as the integrity and credibility of the research itself
ethics in research as the disclosure of methods applied during. the study, where the researcher must always maintain an. impartial position, being honest in his intentions and not. giving away the ...
Synopsis:The integrity of research is based on adherence to core values—objectivity, honesty, openness, fairness, accountability, and stewardship. These core values help to ensure that the research enterprise advances knowledge. Integrity in science means planning, proposing, performing, reporting, and reviewing research in accordance with these values. Participants in the research ...
Abstract. Published articles in scientific journals are a key method for knowledge-sharing. Researchers can face the pressures to publish and this can sometimes lead to a breach of ethical values, whether consciously or unconsciously. The prevention of such practices is achieved by the application of strict ethical guidelines applicable to ...
Abstract. Drawing on various national statements on the ethical conduct of research, the codes of ethics of professional associations and international agencies, and ethical guidelines in social research methods textbooks, this paper identifies current principles for ethical research involving humans and discusses their implications for impact assessment practice generally and social impact ...
At Prolific, we believe in making ethical research easy and accessible. The findings from the Fairwork Cloudwork report speak for themselves. Prolific was given the top score out of all competitors for minimum standards of fair work. With over 25,000 researchers in our community, we're leading the way in revolutionizing the research industry.
Research ethics boards review process may threaten core foundational principles. It is necessary a proportionate representation of qualitative researchers on research ethics boards: McMurphy et al. Canada. 2013: V: To summarize research ethics review experiences in a study about the research ethics review process: Online feedback form of ...
What is Ethics in Research & Why is it Important? W by David B. Resnik, J.D., Ph.D. December 1, 2015 The ideas and opinions expressed in this essay are the author's own and do not necessarily represent those of the NIH, NIEHS, or US government. hen most people think of ethics (or morals), they think of rules for distinguishing between right and
Ethical considerations are foundational to the integrity and validity of scholarly work, guiding researchers to conduct studies that are academically sound and morally responsible. Securing ethics approval is critical for group or individual research projects at any academic level. This process ensures the research respects and protects participants' rights, dignity and welfare.
Introduction. Research includes a set of activities in which researchers use various structured methods to contribute to the development of knowledge, whether this knowledge is theoretical, fundamental, or applied (Drolet & Ruest, accepted).University research is carried out in a highly competitive environment that is characterized by ever-increasing demands (i.e., on time, productivity ...
An "empirical turn" in ethics and integrity research is needed. That would include more empirical work on topics like the actual (lack of) importance of moral norms and values in governance, the effects of more and less ethical and integritous governance on output and outcome (good governance?), and the actual effectiveness of the integrity ...
Research on human subjects — for example, the use of fMRI to study alterations to brain connectivity networks in disease — must be approved by ethics committees (in accordance with the ...
In their examination of survey findings, Pierson et al. (Citation 2024) illuminate critical insights into the current composition and philosophical perspectives of the bioethics field.Their study addresses a significant gap in the systematic understanding of bioethicists' backgrounds and perspectives on key issues, highlighting the profound impact that normative ethical frameworks have on ...
This article presents a mapping review of the literature concerning the ethics of artificial intelligence (AI) in health care. The goal of this review is to summarise current debates and identify open questions for future research. Five literature databases were searched to support the following research question: how can the primary ethical risks presented by AI-health be categorised, and ...
Academic journals, archives, and repositories are seeing an increasing number of questionable research papers clearly produced using generative AI. They are often created with widely available, general-purpose AI applications, most likely ChatGPT, and mimic scientific writing. Google Scholar easily locates and lists these questionable papers alongside reputable, quality-controlled research ...